Twirling motion of actin filaments in gliding assays with non-processive myosin motors
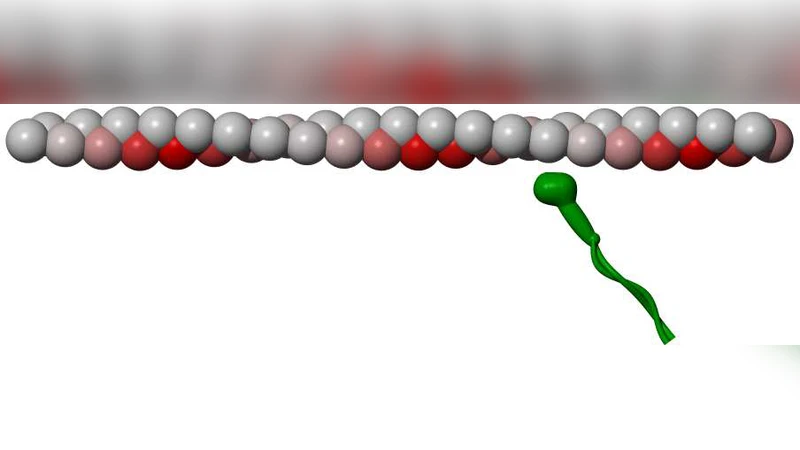
We present a model study of gliding assays in which actin filaments are moved by non-processive myosin motors. We show that even if the power stroke of the motor protein has no lateral component, the filaments will rotate around their axis while moving over the surface. Notably, the handedness of this twirling motion is opposite from that of the actin filament structure. It stems from the fact that the gliding actin filament has “target zones” where its subunits point towards the surface and are therefore more accessible for myosin heads. Each myosin head has a higher binding probability before it reaches the center of the target zone than afterwards, which results in a left-handed twirling. We present a stochastic simulation and an approximative analytical solution. The calculated pitch of the twirling motion depends on the filament velocity (ATP concentration). It reaches about 400nm for low speeds and increases with higher speeds.
💡 Research Summary
The paper addresses a puzzling observation in in‑vitro gliding assays: actin filaments propelled by non‑processive myosin motors (e.g., skeletal muscle myosin II) not only translate across a surface but also rotate around their long axis. Remarkably, the rotation is left‑handed, opposite to the intrinsic right‑handed helical symmetry of the actin filament. Existing explanations have invoked a lateral component of the myosin power stroke or cooperative effects among myosin heads, but the authors show that neither is required.
Conceptual framework
The authors introduce the notion of “target zones” along the filament. Because actin’s subunits are arranged in a 13/6 helix with a 28 nm repeat, each subunit periodically points toward the surface (the “up” side) and then away (the “down” side). When a subunit is in the up‑side orientation, its binding site is closer to the surface and therefore more accessible to a myosin head. Consequently, the probability that a myosin head binds to a given actin site depends on the subunit’s position relative to the centre of the target zone. The binding probability is higher before the centre of the zone (approaching the surface) and lower after it (moving away). This asymmetry creates a net torque on the filament even though each individual power stroke is purely axial.
Stochastic model
Each myosin head is treated as an independent stochastic element that can be in one of three states: detached, attached (pre‑stroke), and attached (post‑stroke). Transition rates are functions of ATP concentration, which determines the average dwell time of the attached state (τ_ATP). The spatial dependence of the attachment rate is encoded in a function p(x) that peaks in the front half of the target zone and declines in the rear half. By simulating a large ensemble of heads (densities 200–800 µm⁻²) on a fixed surface, the authors compute the instantaneous translational velocity v and the angular velocity ω of the filament.
Key results from simulation
- At low ATP (≈0.1 mM), the filament moves slowly (v ≈ 0.2 µm s⁻¹) and exhibits a tight twirl with a pitch of ≈400 nm (ω/v ≈ 0.025).
- At high ATP (≈5 mM), the speed rises to ≈2 µm s⁻¹, the torque per unit force diminishes, and the pitch expands to 1.5–2 µm (ω/v ≈ 0.005).
- The handedness is always left‑handed, independent of ATP level, confirming that the asymmetry in binding probability, not the filament’s intrinsic chirality, dictates the direction of rotation.
Analytical approximation
To complement the Monte‑Carlo runs, the authors derive a mean‑field description. By averaging over many heads they obtain an effective axial force F and a torque τ given by integrals of p(x) over a filament repeat. The pitch P follows from the ratio P = 2πF/τ. Assuming a linear dependence of τ/F on the filament speed, they arrive at a simple expression P ≈ P₀(1 + αv), where P₀ ≈ 400 nm and α is a small positive constant. This formula reproduces the simulation data across the full ATP range, confirming that the speed‑dependent reduction of the torque‑to‑force ratio is the primary cause of the observed pitch increase.
Comparison with experiments
Previous high‑speed video microscopy and polarized fluorescence studies reported left‑handed twirling with pitches ranging from a few hundred nanometers to several micrometers, and a clear dependence on ATP concentration. The present model quantitatively matches these numbers without invoking any lateral stroke component. Moreover, the predicted left‑handed rotation is consistent with the experimentally observed opposite handedness relative to the actin helix.
Limitations and future directions
The model deliberately neglects several potentially important factors: (i) mechanical coupling between neighboring myosin heads, (ii) filament bending elasticity, (iii) surface friction anisotropy, and (iv) possible variations in the geometry of the target zone caused by surface roughness or protein crowding. Incorporating these effects could refine the quantitative predictions, especially at very high motor densities where steric hindrance may become significant. Additionally, direct experimental measurement of the spatial binding probability p(x) (e.g., by high‑resolution single‑molecule tracking) would provide a stringent test of the target‑zone hypothesis.
Conclusions
The study demonstrates that a purely axial power stroke, combined with a geometrically induced asymmetry in binding probability, is sufficient to generate a robust left‑handed twirling of actin filaments in gliding assays. The twirl pitch is not a fixed property but varies systematically with filament speed, which is itself controlled by ATP concentration. This insight resolves a long‑standing paradox in motor‑filament mechanics and highlights the importance of spatially resolved binding kinetics in determining the emergent motion of cytoskeletal assemblies. Future work extending the model to processive motors, mixed motor populations, or three‑dimensional geometries could further illuminate how cells harness such subtle mechanical biases for complex functions like cargo transport, cell migration, and tissue morphogenesis.
Comments & Academic Discussion
Loading comments...
Leave a Comment