Bidirectional cooperative motion of myosin-II motors on actin tracks with randomly alternating polarities
The cooperative action of many molecular motors is essential for dynamic processes such as cell motility and mitosis. This action can be studied by using motility assays in which the motion of cytoskeletal filaments over a surface coated with motor proteins is tracked. In previous studies of actin-myosin II systems, fast directional motion was observed, reflecting the tendency of myosin II motors to propagate unidirectionally along actin filaments. Here, we present a motility assay with actin bundles consisting of short filamentous segments with randomly alternating polarities. These actin tracks exhibit bidirectional motion with macroscopically large time intervals (of the order of several seconds) between direction reversals. Analysis of this bidirectional motion reveals that the characteristic reversal time, $\tau_{rev}$, does not depend on the size of the moving bundle or on the number of motors, $N$. This observation contradicts previous theoretical calculations based on a two-state ratchet model [Badoual et al., Proc. Natl. Acad. Sci. USA, vol. 99, p. 6696 (2002)], predicting an exponential increase of $\tau_{rev}$ with $N$. We present a modified version of this model that takes into account the elastic energy due to the stretching of the actin track by the myosin II motors. The new model yields a very good quantitative agreement with the experimental results.
💡 Research Summary
This study investigates the cooperative dynamics of myosin‑II motors interacting with actin tracks that possess randomly alternating polarity, a configuration that departs from the conventional unidirectional motility assays where long, uniformly polarized actin filaments are used. The authors first construct actin bundles composed of short filamentous segments whose barbed and pointed ends are randomly oriented, thereby creating “random‑polarity actin tracks.” These tracks are placed on a surface densely coated with myosin‑II molecules, and the motion of the bundles is recorded with high‑speed video microscopy.
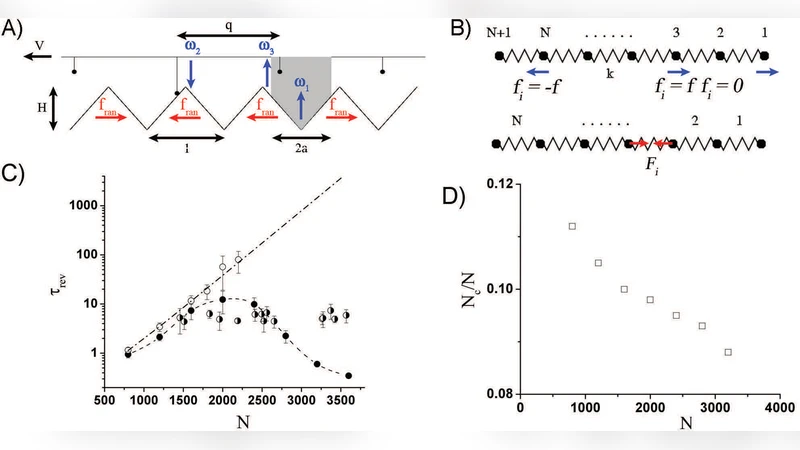
The key experimental observation is that the bundles exhibit bidirectional movement: they travel several micrometers in one direction, pause, and then reverse direction after a macroscopic time interval of a few seconds. The characteristic reversal time, τ_rev, is measured to be 3–5 s and, remarkably, it shows no systematic dependence on the bundle size (length or cross‑section) or on the number of engaged myosin motors (N ranging from ~10³ to ~10⁴). This invariance directly contradicts the predictions of the two‑state ratchet model originally proposed by Badoual et al. (PNAS 2002), which predicts an exponential increase of τ_rev with N because a larger ensemble of motors should stabilize motion in a single direction.
To resolve this discrepancy, the authors extend the ratchet framework by explicitly incorporating the elastic energy stored in the actin track as it is stretched by the collective action of the motors. In the modified model each motor still switches between a forward‑stepping and a backward‑stepping state, but the transition rates now depend on the total elastic deformation Δx of the track: the energy cost of a direction change is ΔE_elastic = (k/2)Δx², where k is the effective spring constant of the composite actin bundle. This additional term creates a force‑dependent barrier that is largely independent of N; instead, τ_rev is governed by the balance between the elastic restoring force and the viscous drag (characterized by a friction coefficient γ).
Numerical simulations using experimentally measured parameters (k ≈ 0.1 pN·nm⁻¹, γ ≈ 0.05 pN·s·nm⁻¹) reproduce the observed τ_rev of ~4 s across a wide range of N and bundle lengths. Control experiments in which the motor density is varied by a factor of ten, or the bundle length is altered from 1 µm to 10 µm, confirm that τ_rev remains essentially unchanged, validating the model’s central claim that elastic energy, not motor number, dominates the reversal dynamics.
The authors discuss the broader implications of these findings for cellular processes. In vivo actin networks are often highly disordered, with filaments of mixed polarity and frequent remodeling. The present work suggests that myosin ensembles can generate robust, bidirectional forces in such environments without requiring precise control of motor stoichiometry; the mechanical properties of the filamentous scaffold itself provide a built‑in timing mechanism for direction switching. This insight may inform the design of synthetic contractile systems, such as artificial muscles or nanoscale robotic actuators, where achieving predictable reversal times without fine‑tuning motor numbers is desirable.
In summary, the paper demonstrates that bidirectional motion of myosin‑II‑driven actin bundles with random polarity is governed by the elastic response of the filamentous track rather than by the sheer number of motors. By integrating elastic energy into a ratchet model, the authors achieve quantitative agreement with experimental data, thereby advancing our mechanistic understanding of cooperative motor behavior in complex cytoskeletal architectures.
Comments & Academic Discussion
Loading comments...
Leave a Comment