Lipid Domain Order and the Algebra of Morphology
Lipid membranes regulate the flow of materials and information between cells and their organelles. Further, lipid composition and morphology can play a key role in regulating a variety of biological processes. For example, viral uptake, plasma membrane tension regulation, and the formation of caveolae all require the creation and control of groups of lipids that adopt specific morphologies. In this paper, we use a simplified model mixture of lipids and cholesterol to examine the interplay between lipid phase-separation and bilayer morphology. We observe and theoretically analyze three main features of phase-separated giant unilamellar vesicles. First, by tracking the motion of dimpled' domains, we measure repulsive, elastic interactions that create short--range translational and orientational order, leading to a stable distribution of domain sizes, and hence maintaining lateral heterogeneity on relatively short length scales and long time scales. Second, we examine the transition to budded’ domain morphologies, showing that the transition is size-selective, and has two kinetic regimes, as revealed by a calculated phase diagram. Finally, using observations of the interactions between dimpled and budded domains, we build a theoretical framework with an elastic model that maps the free energies and allowed transitions in domain morphology upon coalescence, to serve as an interpretive tool for understanding the algebra of domain morphology. In all three cases, the two major factors that regulate domain morphology and morphological transitions are the domain size and membrane tension.
💡 Research Summary
This paper investigates how lipid composition, phase separation, and membrane tension together dictate the morphology of domains in model lipid‑cholesterol membranes. Using giant unilamellar vesicles (GUVs) composed of a simplified ternary mixture, the authors combine high‑resolution fluorescence microscopy, particle‑tracking algorithms, and controlled aspiration to vary membrane tension while monitoring domain behavior over time.
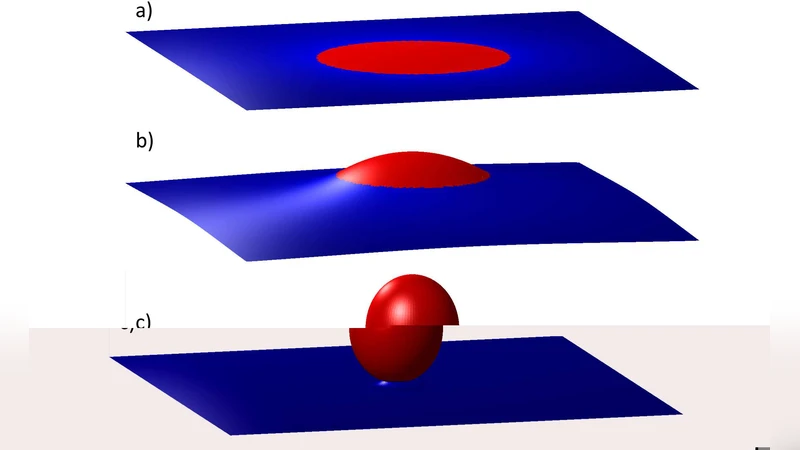
The first major observation concerns “dimpled” domains—regions where the bilayer locally indents, forming a shallow depression. By tracking the trajectories of many such domains, the authors quantify a short‑range repulsive elastic interaction that depends strongly on inter‑domain distance. When two dimples approach within roughly two to three times their radius, the repulsive force rises sharply, leading to a self‑organized arrangement characterized by translational and orientational order. This ordering limits the spread of domain sizes, producing a relatively narrow, stable size distribution that persists over long timescales, thereby maintaining lateral heterogeneity in the membrane. The authors model this behavior using an elastic energy framework that balances bending rigidity, membrane tension, and line tension at the domain boundary, showing that the observed spacing corresponds to a minimum‑energy configuration for a given domain density.
The second focus is the transition from dimpled to “budded” domains. Budding occurs when a domain exceeds a critical size, causing the membrane to protrude outward and form a spherical cap connected by a narrow neck. The critical size is not fixed; it shifts with membrane tension. Low tension permits budding at smaller radii, whereas high tension suppresses the transition, requiring larger domains. Kinetic analysis reveals two distinct regimes: an initial rapid “nucleation” phase where the bud forms, followed by a slower “relaxation” phase during which the neck geometry adjusts and the surrounding membrane redistributes stress. By solving the coupled equations for bending energy, tension, and line tension, the authors construct a phase diagram in the tension‑size plane that delineates the boundaries between flat, dimpled, and budded states.
The third contribution integrates the interactions between dimpled and budded domains. When a dimpled domain collides with a bud, the system can undergo coalescence, leading to a change in overall morphology. To rationalize these events, the authors develop a comprehensive elastic free‑energy model:
F = ∫
Comments & Academic Discussion
Loading comments...
Leave a Comment