Synchronized Dynamics and Nonequilibrium Steady States in a Stochastic Yeast Cell-Cycle Network
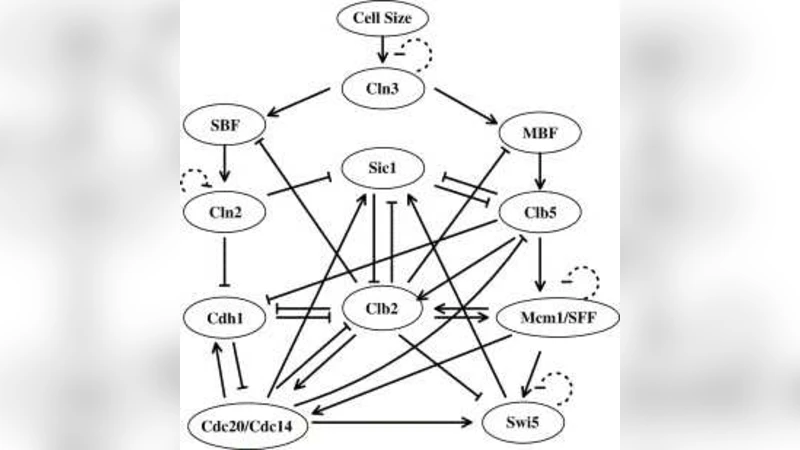
Applying the mathematical circulation theory of Markov chains, we investigate the synchronized stochastic dynamics of a discrete network model of yeast cell-cycle regulation where stochasticity has been kept rather than being averaged out. By comparing the network dynamics of the stochastic model with its corresponding deterministic network counterpart, we show that the synchronized dynamics can be soundly characterized by a dominant circulation in the stochastic model, which is the natural generalization of the deterministic limit cycle in the deterministic system. Moreover, the period of the main peak in the power spectrum, which is in common use to characterize the synchronized dynamics, perfectly corresponds to the number of states in the main cycle with dominant circulation. Such a large separation in the magnitude of the circulations, between a dominant, main cycle and the rest, gives rise to the stochastic synchronization phenomenon.
💡 Research Summary
The paper applies the circulation theory of Markov chains to a stochastic, discrete‑time model of the yeast cell‑cycle regulatory network, preserving intrinsic noise rather than averaging it out. Starting from the well‑known deterministic Boolean network that exhibits a 13‑state limit cycle, the authors construct a stochastic counterpart by assigning transition probabilities to each node based on a logistic function with a noise parameter ε. This yields a finite‑state Markov chain (2¹¹ = 2048 states) whose transition matrix is fully positive.
Using the cycle‑decomposition theorem, the overall probability flow is expressed as a weighted sum of elementary cycles Cₖ, each characterized by a circulation γ(Cₖ), i.e., the product of transition probabilities along the cycle. Numerical computation of all circulations reveals a striking hierarchy: one particular cycle—identical to the deterministic limit cycle—carries a circulation orders of magnitude larger than any other cycle. This “dominant circulation” therefore governs the long‑time behavior of the stochastic system.
A power‑spectral analysis of the time series generated by Monte‑Carlo simulations shows a prominent peak whose period equals the length of the dominant cycle (13 steps). The perfect match between the spectral peak and the number of states in the dominant cycle demonstrates that the usual experimental indicator of synchronization (the main peak in the power spectrum) directly reflects the underlying dominant circulation.
The authors further explore how the separation between the dominant circulation and the rest of the circulations depends on ε. When ε is small (weak noise), the dominant circulation remains overwhelmingly large, and the system behaves almost deterministically, reproducing the classic limit‑cycle dynamics. As ε increases, the circulations of non‑dominant cycles grow, the hierarchy collapses, and the spectral peak disappears, indicating loss of synchronization. This transition is interpreted as a shift from a non‑equilibrium steady state with highly asymmetric probability flux (large entropy production concentrated in the dominant cycle) to a more symmetric flux distribution where noise dominates.
Key contributions of the work are:
- Methodological Innovation – Introducing Markov‑chain circulation analysis as a rigorous tool to quantify stochastic synchronization in discrete biological networks.
- Theoretical Bridge – Demonstrating that the deterministic limit cycle is the zero‑noise limit of a dominant stochastic circulation, thereby unifying deterministic and stochastic descriptions.
- Empirical Link – Connecting the dominant circulation to the main peak of the power spectrum, providing a clear interpretation for experimental time‑series data.
- Noise‑Induced Transition – Showing that the magnitude gap between circulations controls the onset or loss of synchronization, offering insight into how cellular noise can modulate rhythmic processes.
Finally, the authors argue that the same framework can be applied to other oscillatory biological systems (e.g., circadian clocks, p53 pulses), where stochastic effects are significant. By quantifying the hierarchy of circulations, researchers can predict whether a noisy network will exhibit coherent rhythmic behavior or devolve into random fluctuations. The study thus advances our understanding of how robust biological rhythms emerge and persist in the presence of molecular noise.
Comments & Academic Discussion
Loading comments...
Leave a Comment