Non-equilibrium phase transitions in tubulation by molecular motors
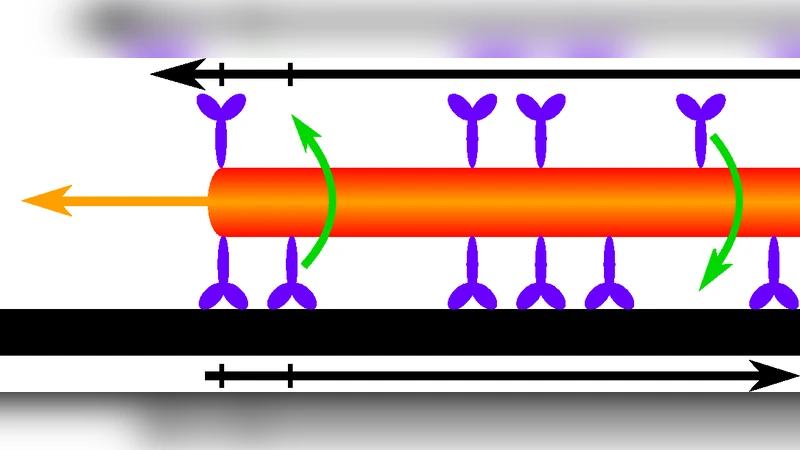
The extraction of membrane tubes by molecular motors is known to play an important role for the transport properties of eukaryotic cells. By studying a generic class of models for the tube extraction, we discover a rich phase diagram. In particular we show that the density of motors along the tube can exhibit shocks, inverse shocks and plateaux, depending on parameters which could in principle be probed experimentally. In addition the phase diagram exhibits interesting reentrant behavior.
💡 Research Summary
The paper investigates how ensembles of molecular motors extract membrane tubes—a process known as tubulation—that is crucial for intracellular transport. The authors construct a generic class of non‑equilibrium lattice models that extend the asymmetric simple exclusion process (ASEP) by incorporating motor attachment/detachment at the tube ends and two internal states corresponding to forward and backward stepping. The key parameters are the entry rate α (motor binding at the tube base), the exit rate β (motor unbinding at the tip), and the forward/backward hopping rates p and q, which can be tuned experimentally through ATP concentration, motor type, or membrane tension.
Using mean‑field analysis together with extensive Monte‑Carlo simulations, the authors derive the steady‑state density profile ρ(x) along the tube and the associated current J(ρ)=pρ(1−ρ)−qρ(1−ρ). Boundary conditions imposed by α and β generate two possible bulk fixed points, identified as low‑density (LD) and high‑density (HD) phases. The competition between these fixed points gives rise to a rich phase diagram.
The most striking findings are:
-
Shocks – For a broad range of α/β ratios a sharp discontinuity (shock) separates an LD region near the base from an HD region near the tip. The shock position moves continuously with the parameters and can be directly linked to motor accumulation or depletion zones.
-
Inverse shocks – When the forward and backward stepping rates are highly asymmetric (p≫q or q≫p), the shock can reverse its orientation, producing an HD region at the base and an LD region at the tip. This “inverse shock” is absent in the standard ASEP and reflects the non‑trivial coupling between motor stepping bias and boundary fluxes.
-
Plateaux – In a distinct parameter window the density profile becomes flat over a macroscopic segment of the tube, forming a plateau. In this regime the current is saturated and the tube maintains a uniform tension and radius, suggesting a mechanically stable configuration.
-
Re‑entrant behavior – Varying a single control parameter (e.g., α) can drive the system through a sequence of phases: LD → shock → plateau → inverse shock → LD again. Consequently, the same α value may correspond to different macroscopic states, a hallmark of non‑linear non‑equilibrium systems.
The authors discuss how each of these phenomena could be probed experimentally. Fluorescently labeled motors allow real‑time measurement of the longitudinal density, while high‑speed cylindrical microscopy can track tube length and radius. By adjusting ATP levels (modulating p and q) or motor concentration (changing α and β), one can deliberately induce shocks, inverse shocks, or plateaux and verify the predicted re‑entrant transitions.
Beyond the specific biological context, the work demonstrates how extending ASEP to include attachment/detachment and bidirectional stepping yields a versatile framework for describing active transport on deformable substrates. The identified non‑equilibrium phase transitions provide a mechanistic explanation for how cells might dynamically regulate tube formation in response to environmental cues, and they offer design principles for synthetic nanodevices that harness motor‑driven membrane remodeling.
Comments & Academic Discussion
Loading comments...
Leave a Comment