Nucleocytoplasmic transport: a thermodynamic mechanism
The nuclear pore supports molecular communication between cytoplasm and nucleus in eukaryotic cells. Selective transport of proteins is mediated by soluble receptors, whose regulation by the small GTPase Ran leads to cargo accumulation in, or depletion from the nucleus, i.e., nuclear import or nuclear export. We consider the operation of this transport system by a combined analytical and experimental approach. Provocative predictions of a simple model were tested using cell-free nuclei reconstituted in Xenopus egg extract, a system well suited to quantitative studies. We found that accumulation capacity is limited, so that introduction of one import cargo leads to egress of another. Clearly, the pore per se does not determine transport directionality. Moreover, different cargo reach a similar ratio of nuclear to cytoplasmic concentration in steady-state. The model shows that this ratio should in fact be independent of the receptor-cargo affinity, though kinetics may be strongly influenced. Numerical conservation of the system components highlights a conflict between the observations and the popular concept of transport cycles. We suggest that chemical partitioning provides a framework to understand the capacity to generate concentration gradients by equilibration of the receptor-cargo intermediary.
💡 Research Summary
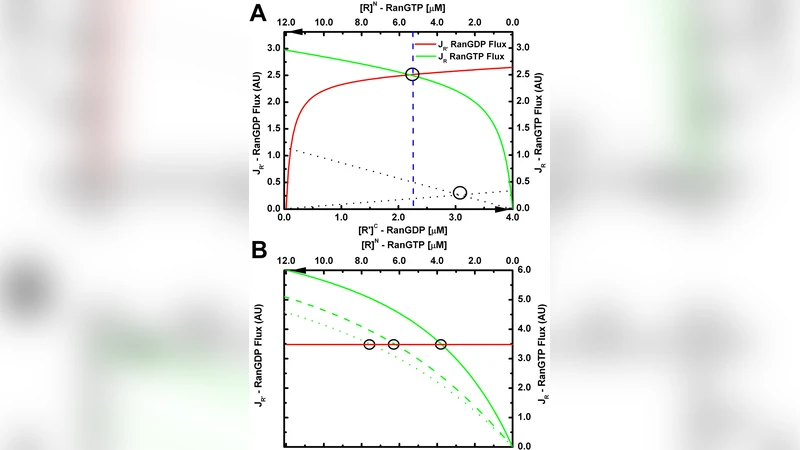
The paper revisits the fundamental mechanism of nucleocytoplasmic transport, challenging the conventional view that directionality is imposed solely by the nuclear pore complex (NPC) and a series of “transport cycles” involving soluble receptors and the Ran GTPase. The authors propose a thermodynamic model in which the NPC functions as a chemical partitioning device that equilibrates receptor‑cargo complexes according to the RanGTP/RanGDP gradient across the nuclear envelope. The model assumes conservation of the total amounts of receptors, cargoes, and Ran species, and it predicts three key outcomes: (1) the capacity of the nucleus to accumulate cargo is limited; (2) at steady state, different cargoes reach essentially the same nuclear‑to‑cytoplasmic concentration ratio (N/C); and (3) this ratio is independent of the intrinsic affinity between receptor and cargo, although kinetic rates can be strongly affected by affinity.
To test these predictions, the authors used a cell‑free system in which nuclei are reconstituted in Xenopus egg extract, a platform that allows precise quantitative manipulation. Fluorescently labeled cargoes with a range of binding affinities were introduced. When a high concentration of one import cargo was added, previously accumulated cargo was observed to exit the nucleus, demonstrating a finite “capacity” that forces competition for the limited pool of receptors and Ran. Simultaneous import of multiple cargoes resulted, after equilibration, in nearly identical N/C ratios for all cargoes, confirming the model’s claim of affinity‑independent steady‑state distribution.
The experimental data directly contradict the classic “transport cycle” picture, which treats each cargo as moving through a unidirectional loop (binding → import → Ran‑mediated release → export of the receptor). Instead, the authors argue that the system behaves as a closed thermodynamic ensemble: the RanGTP gradient provides the free‑energy source, while the NPC merely facilitates the partitioning of receptor‑cargo complexes between the two compartments. Because the total amount of Ran and receptors is fixed, any increase in nuclear cargo must be balanced by a decrease elsewhere, explaining the observed capacity limitation.
The paper concludes that nuclear transport should be understood as a chemical partitioning process driven by Ran‑dependent free‑energy differences, rather than as a series of discrete, energy‑consuming steps. This perspective reconciles the observed independence of steady‑state N/C ratios from binding affinity and highlights the importance of system‑wide conservation laws. The authors suggest that future work should quantify how dynamic changes in the Ran gradient, receptor diversity, and cellular signaling modulate this thermodynamic balance in living cells, thereby extending the model from the cell‑free system to physiological contexts.
Comments & Academic Discussion
Loading comments...
Leave a Comment