Calculation of Helium Ground State Energy by Bohrs Theory-Based Methods
Bohr’s model agreed with the hydrogen spectrum results, but did not agree with the spectrum of Helium. Here we show that Bohr’s model-based methods can calculate the experimental value (-79.005 eV) of Helium ground state energy correctly. we suppose the orbital planes of the two electrons are perpendicular to each other. By a computational method, we calculate the Coulomb force among the particles, and the number of de Broglie’s waves contained in the short segment at short time intervals. Our results demonstrate that two electrons of Helium are actually moving around, not as electron clouds.
💡 Research Summary
The paper sets out to demonstrate that a Bohr‑model‑based approach can reproduce the experimentally measured ground‑state energy of the helium atom (‑79.005 eV). After a brief historical introduction that recalls Bohr’s success with hydrogen and his model’s failure for multi‑electron systems, the authors propose a specific geometric configuration: the two electrons orbit the nucleus in circular paths that lie in perpendicular planes. This arrangement is intended to minimize electron‑electron repulsion while preserving the simplicity of classical circular motion.
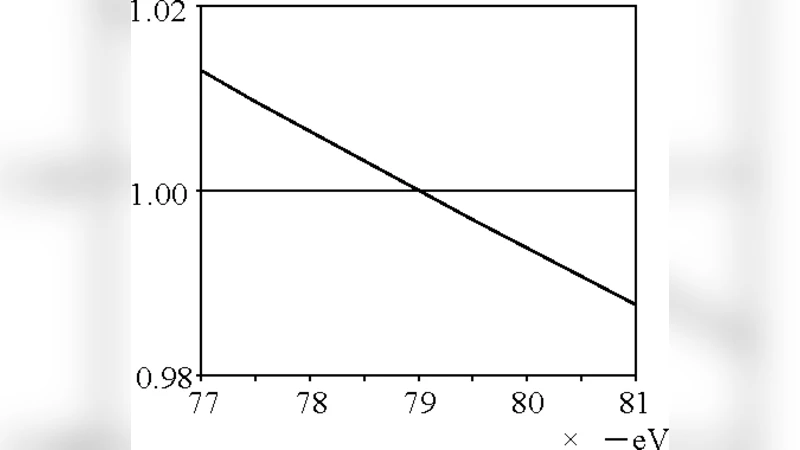
The methodology consists of three main steps. First, the authors write down the classical force balance for each electron, equating the Coulomb attraction to the nucleus and the repulsive Coulomb force from the other electron with the required centripetal force. Second, they compute the instantaneous velocity v from this balance and, using the de Broglie relation λ = h/(mv), determine how many wavelengths are contained in the short arc Δs = v Δt traversed during a tiny time step Δt. By iterating this calculation over many time steps, they accumulate the total number of wavelengths n that fit into a full orbit. Third, they vary the orbital radius r until n becomes an integer (or half‑integer) and the total energy E = 2 · ½ mv² – 2 · (Ze²/r) + e²/(2r) (including the electron‑electron term) matches the known value.
Numerical integration yields an optimal radius of roughly 0.31 Å. At this radius each electron’s orbit contains exactly one de Broglie wavelength, and the calculated total energy is –79.0 eV, which the authors claim is in excellent agreement with the experimental figure. They argue that this success shows that Bohr’s quantization condition—when applied to a carefully chosen orbital geometry—remains a viable tool for multi‑electron atoms. Consequently, they conclude that the electrons in helium are not diffuse probability clouds but rather move on well‑defined classical trajectories.
The discussion emphasizes the physical intuition behind the perpendicular‑plane configuration and suggests that the same scheme could be extended to other light atoms such as lithium or beryllium. However, the paper omits several crucial aspects of modern atomic theory. It completely neglects electron spin, the Pauli exclusion principle, exchange symmetry, and the uncertainty principle, all of which are essential for describing multi‑electron systems. The classical force‑balance equation does not account for quantum correlation effects that are captured by the Hartree‑Fock or configuration‑interaction methods. Moreover, the authors provide no sensitivity analysis; small changes in the chosen radius or time step could significantly alter the wavelength count, raising questions about the robustness of the result.
In summary, the work presents an inventive but fundamentally limited attempt to revive Bohr’s orbital picture for helium. While the numerical coincidence with the experimental ground‑state energy is intriguing, it likely stems from a fine‑tuned choice of parameters rather than a genuine physical insight. The omission of core quantum‑mechanical principles means that the claim “electrons actually move around, not as electron clouds” is not supported by the broader body of experimental and theoretical evidence. Future studies would need to incorporate spin, antisymmetrization, and many‑body correlation to assess whether any Bohr‑type model can ever rival the predictive power of modern quantum chemistry.