Modeling thalamocortical cell: impact of Ca2+ channel distribution and cell geometry on firing pattern
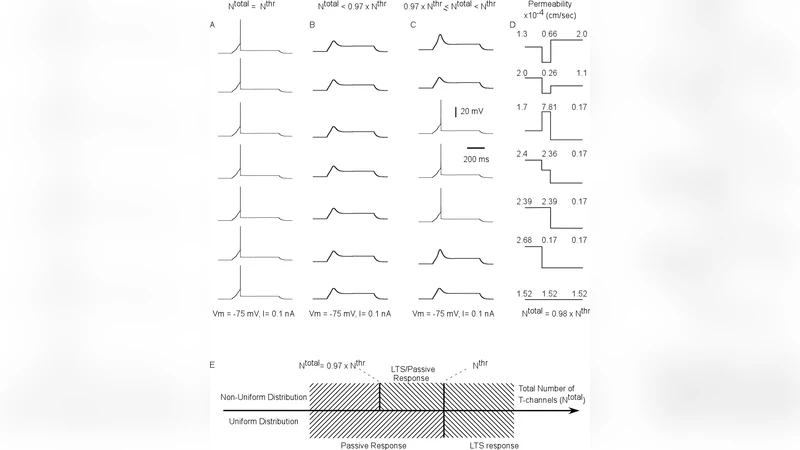
The influence of calcium channel distribution and geometry of the thalamocortical cell upon its tonic firing and the low threshold spike (LTS) generation was studied in a 3-compartment model, which represents soma, proximal and distal dendrites as well as in multi-compartment model using the morphology of a real reconstructed neuron. Using an uniform distribution of Ca2+ channels, we determined the minimal number of low threshold voltage-activated calcium channels and their permeability required for the onset of LTS in response to a hyperpolarizing current pulse. In the 3-compartment model, we found that the channel distribution influences the firing pattern only in the range of 3% below the threshold value of total T-channel density. In the multi-compartmental model, the LTS could be generated by only 64% of unequally distributed T-channels compared to the minimal number of equally distributed T-channels. For a given channel density and injected current, the tonic firing frequency was found to be inversely proportional to the size of the cell. However, when the Ca2+ channel density was elevated in soma or proximal dendrites, then the amplitude of LTS response and burst spike frequencies were determined by the ratio of total to threshold number of T-channels in the cell for a specific geometry.
💡 Research Summary
The paper investigates how the spatial distribution of low‑threshold voltage‑activated calcium (T‑type) channels and the geometric properties of thalamocortical (TC) neurons shape their firing behavior, focusing on tonic firing and low‑threshold spikes (LTS). Two computational frameworks are employed: a simplified three‑compartment model (soma, proximal dendrite, distal dendrite) and a detailed multi‑compartment model built from a reconstructed TC neuron morphology.
In the three‑compartment model, the authors first determine the minimal number of T‑channels and the associated permeability required to generate an LTS when the cell receives a hyperpolarizing current pulse. By systematically varying the total channel density, they find that the distribution of channels (whether uniform or slightly non‑uniform) only influences the firing pattern when the total channel density lies within a narrow window—approximately 3 % below the threshold value needed for LTS initiation. Outside this narrow band, the firing pattern is essentially invariant to the exact placement of channels, indicating that once a critical pool of T‑channels is present, the precise spatial arrangement becomes less critical.
The multi‑compartment model incorporates realistic dendritic branching, varying diameters, and electrotonic length constants. When T‑channels are distributed non‑uniformly—concentrated in the soma and proximal dendrites—the model can evoke an LTS with only 64 % of the total channels required in the uniformly distributed case. This striking reduction demonstrates that dendritic geometry creates “hot spots” where channel density is amplified electrotonically, allowing fewer channels to produce the same depolarizing drive. The authors quantify this effect by introducing a ratio of total channels to the threshold number of channels (the “total‑to‑threshold ratio”). For a given geometry, the amplitude of the LTS and the frequency of burst spikes scale with this ratio rather than with absolute channel count.
A second major finding concerns tonic firing. Holding channel density and injected current constant, the authors show that the tonic firing frequency is inversely proportional to cell size. Larger cells have lower input resistance and larger capacitance, which dampens voltage excursions and slows the inter‑spike interval. Conversely, smaller cells fire more rapidly under identical conditions. This relationship holds across both model types, underscoring the fundamental role of passive membrane properties in setting firing rates.
When the density of T‑channels is selectively increased in the soma or proximal dendrites, the model predicts two synergistic effects: (1) the LTS becomes larger in amplitude, and (2) the subsequent burst of high‑frequency spikes is more robust. Importantly, these effects are not linear with channel number; instead, they depend on how the total channel count compares to the threshold count for that specific geometry. Thus, a cell with a high total‑to‑threshold ratio will exhibit pronounced LTS and burst firing, whereas a cell near the threshold will produce only a modest depolarizing hump.
The study provides several conceptual insights. First, the threshold number of T‑channels is a critical determinant of whether a TC neuron can switch from tonic to burst mode. Second, dendritic architecture can dramatically lower the effective threshold by concentrating channels in electrotonically privileged regions. Third, passive properties (cell size, membrane resistance, capacitance) modulate tonic firing independently of active channel distribution. Fourth, the total‑to‑threshold ratio offers a compact metric to predict LTS amplitude and burst characteristics across diverse morphologies.
From a physiological perspective, these results help explain why thalamic relay neurons display heterogeneous firing patterns despite sharing similar channel complements. Variations in dendritic branching, soma size, and channel localization can shift a neuron’s operating point along the tonic‑burst continuum, influencing sensory gating, sleep rhythms, and thalamocortical oscillations. Clinically, the framework could be applied to pathological conditions where T‑channel expression or dendritic structure is altered, such as absence epilepsy or thalamic stroke, offering predictions about altered burst propensity and potential therapeutic targets.
Overall, the paper combines systematic parameter sweeps with biologically realistic morphologies to dissect the interplay between channel distribution, cell geometry, and firing dynamics, delivering a quantitative foundation for future experimental and modeling work on thalamic function.
Comments & Academic Discussion
Loading comments...
Leave a Comment