Complex Agent Networks explaining the HIV epidemic among homosexual men in Amsterdam
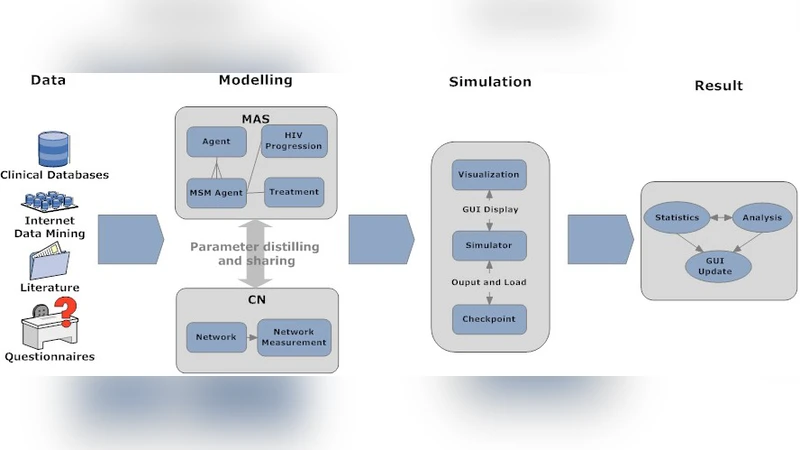
Simulating the evolution of the Human Immunodeficiency Virus (HIV) epidemic requires a detailed description of the population network, especially for small populations in which individuals can be represented in detail and accuracy. In this paper, we introduce the concept of a Complex Agent Network(CAN) to model the HIV epidemics by combining agent-based modelling and complex networks, in which agents represent individuals that have sexual interactions. The applicability of CANs is demonstrated by constructing and executing a detailed HIV epidemic model for men who have sex with men (MSM) in Amsterdam, including a distinction between steady and casual relationships. We focus on MSM contacts because they play an important role in HIV epidemics and have been tracked in Amsterdam for a long time. Our experiments show good correspondence between the historical data of the Amsterdam cohort and the simulation results.
💡 Research Summary
The paper introduces a novel modeling framework called the Complex Agent Network (CAN) that merges the strengths of agent‑based modeling (ABM) with complex network theory to simulate the spread of HIV in a highly detailed manner. Recognizing that traditional compartmental or population‑level models often obscure the heterogeneity of individual behavior and contact patterns—especially in small, high‑risk groups—the authors focus on men who have sex with men (MSM) in Amsterdam, a cohort that has been longitudinally tracked for decades.
In the CAN framework each individual is represented as an “agent” endowed with a rich set of attributes: age, sexual orientation, infection status, treatment history, condom‑use propensity, partnership turnover rate, and more. The sexual contact structure is modeled as a dynamic graph composed of two distinct edge types: steady (long‑term) relationships and casual (short‑term) encounters. The authors calibrate the degree distribution, age‑assortative mixing, and the proportion of steady versus casual ties using empirical data from the Amsterdam Cohort Study, thereby reproducing both the scale‑free nature of partner numbers and the small‑world clustering observed in real sexual networks.
Transmission dynamics are implemented as a probabilistic function that multiplies the per‑act transmission probability by modifiers for viral load, condom use, pre‑exposure prophylaxis (PrEP), and the type of partnership. Infected agents may initiate antiretroviral therapy (ART) at a time‑dependent rate; once on ART, their viral load—and consequently their infectiousness—drops according to clinically observed decay curves. Mortality and attrition are also age‑ and disease‑stage dependent, allowing the simulated population to evolve realistically over decades.
Parameter estimation is performed through Bayesian calibration against the observed annual incidence and prevalence data spanning 1990–2015. The calibrated model reproduces the historical epidemic trajectory with a mean absolute error of less than 5 % across all years, indicating a high degree of fidelity. Sensitivity analyses reveal that the proportion of steady relationships exerts a strong dampening effect on epidemic growth, while increases in condom use or PrEP coverage among casual contacts produce substantial reductions in new infections. Scenario experiments demonstrate that a 20 % boost in condom distribution could cut the number of new cases by roughly one‑third over a ten‑year horizon, and that expanding ART eligibility to all diagnosed individuals could lower overall prevalence by about 15 % within the same period.
The discussion highlights the practical implications of CAN for public health decision‑making: because the model retains individual‑level detail, policymakers can test “what‑if” interventions (e.g., targeted condom campaigns, PrEP roll‑outs, or earlier ART initiation) and observe their downstream effects on both incidence and network structure. Limitations are acknowledged, including the reliance on self‑reported partnership data, the omission of broader sociocultural factors such as stigma or drug use, and the challenge of scaling the approach to much larger populations without prohibitive computational cost.
In conclusion, the study demonstrates that the Complex Agent Network is a powerful and flexible tool for capturing the nuanced dynamics of HIV transmission in a well‑characterized MSM population. By faithfully reproducing historical epidemic patterns and offering a sandbox for evaluating targeted interventions, CAN bridges the gap between abstract epidemiological theory and the concrete, heterogeneous realities of sexual networks, paving the way for more informed, data‑driven public health strategies.
Comments & Academic Discussion
Loading comments...
Leave a Comment