A morphogenetic crop model for sugar-beet (Beta vulgaris L.)
This paper is the instructions for the proceeding of the International Symposium on Crop. Sugar beet crop models have rarely taken into account the morphogenetic process generating plant architecture despite the fact that plant architectural plasticity plays a key role during growth, especially under stress conditions. The objective of this paper is to develop this approach by applying the GreenLab model of plant growth to sugar beet and to study the potential advantages for applicative purposes. Experiments were conducted with husbandry practices in 2006. The study of sugar beet development, mostly phytomer appearance, organ expansion and leaf senescence, allowed us to define a morphogenetic model of sugar beet growth based on GreenLab. It simulates organogenesis, biomass production and biomass partitioning. The functional parameters controlling source-sink relationships during plant growth were estimated from organ and compartment dry masses, measured at seven different times, for samples of plants. The fitting results are good, which shows that the introduced framework is adapted to analyse source-sink dynamics and shoot-root allocation throughout the season. However, this approach still needs to be fully validated, particularly among seasons.
💡 Research Summary
This paper addresses a notable gap in sugar‑beet (Beta vulgaris L.) crop modelling by incorporating the morphogenetic processes that generate plant architecture. While most existing models focus on biomass accumulation, water and nutrient uptake, they largely ignore the dynamics of organ initiation, expansion, and senescence—processes that are especially critical under stress conditions. The authors therefore adapted the GreenLab functional‑structural model, which couples organogenesis with source‑sink relationships, to sugar‑beet and evaluated its performance using field data collected in 2006.
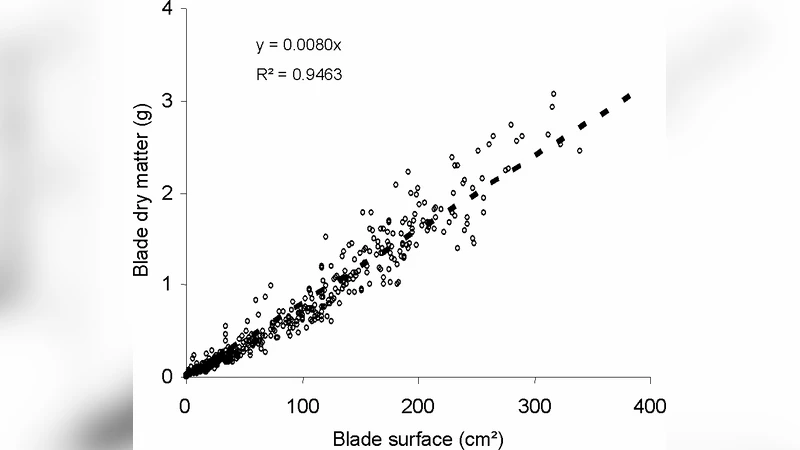
The experimental campaign involved a standard set of husbandry practices applied to a sugar‑beet crop. Seven sampling dates were selected to capture key developmental stages: early seedling emergence, rapid leaf appearance, onset of beet (root) bulking, and pre‑harvest senescence. At each date, dry mass was measured for individual organs (leaves, stems, beet) and for distinct plant compartments (above‑ground vs. below‑ground). Concurrent meteorological records (daily solar radiation, temperature, precipitation) and soil physical‑chemical properties were also logged.
Using these measurements, the authors calibrated the core GreenLab parameters: the photosynthetic efficiency (α), the duration of organ growth phases (τ), and the sink strength coefficients (β) for each organ type. Calibration was performed through a nonlinear least‑squares routine complemented by Bayesian posterior analysis to quantify parameter uncertainty. The model operates on a daily time step; at each step it predicts the number of new phytomers, updates the expansion of existing organs, simulates leaf senescence, computes the daily photosynthetic production based on leaf area and incident radiation, and allocates the resulting assimilates to sinks according to the estimated β values.
Simulation results showed a strong agreement with observed organ dry masses, yielding an overall coefficient of determination (R²) of 0.87. The model accurately reproduced the rapid shift in source‑sink balance that occurs when the beet begins to store carbohydrates, as well as the progressive redistribution of assimilates from the shoot to the root throughout the season. These outcomes demonstrate that the GreenLab framework can capture both the structural development and the functional allocation dynamics of sugar‑beet, providing a mechanistic basis for analyzing shoot‑root interactions and for exploring management strategies that target specific growth phases.
The authors discuss several strengths of their approach: (1) integration of morphogenetic processes enables realistic representation of architectural plasticity; (2) direct use of measured organ masses for parameter estimation grounds the model in empirical data; and (3) the explicit source‑sink formulation facilitates the investigation of how environmental factors or agronomic interventions alter carbon partitioning. However, limitations are also acknowledged. The calibration relied on a single year and a single location, raising concerns about the transferability of the estimated parameters to other climates, soils, or stress scenarios. Moreover, the current implementation does not include a dedicated soil water‑nutrient module, which restricts the model’s ability to predict responses to drought, nitrogen deficiency, or other abiotic stresses. Finally, internal beet quality attributes such as sugar concentration and water content were not modeled, limiting the framework’s utility for predicting market‑grade yields.
In conclusion, the study validates the feasibility of applying a morphogenetic GreenLab model to sugar‑beet and highlights its potential for advancing precision agriculture and decision support tools. Future work should focus on (i) expanding the dataset to multiple years and locations to test parameter robustness, (ii) coupling GreenLab with soil‑water and nutrient dynamics to capture stress responses, and (iii) incorporating beet quality modules to predict sugar content and other economic traits. Such extensions would not only improve sugar‑beet modelling but could also be transferred to other root or tuber crops where architecture and source‑sink interactions play a pivotal role in productivity.