Cell-to-Cell stochastic fluctuations in apoptotic signaling can decide between life and death
Apoptosis, or genetically programmed cell death, is a crucial cellular process that maintains the balance between life and death in cells. The precise molecular mechanism of apoptosis signaling and how these two pathways are differentially activated under distinct apoptotic stimuli is poorly understood. We developed a Monte Carlo-based stochastic simulation model that can characterize distinct signaling behaviors in the two major pathways of apoptotic signaling using a novel probability distribution-based approach. Specifically, we show that for a weak death signal, such as low levels of death ligand Fas (CD95) binding or under stress conditions, the type 2 mitochondrial pathway dominates apoptotic signaling. Our results also show signaling in the type 2 pathway is stochastic, where the population average over many cells does not capture the cell-to-cell fluctuations in the time course (~1 - 10 hours) of downstream caspase-3 activation. On the contrary, the probability distribution of caspase-3 activation for the mitochondrial pathway shows a distinct bimodal behavior that can be used to characterize the stochastic signaling in type 2 apoptosis. Interestingly, such stochastic fluctuations in apoptosis signaling happen even in the presence of large numbers of signaling molecules. In a fluctuating environment, such stochasticity in the timecourse of caspase-3 activation may be an adaptive mechanism for allowing a competing survival signal to win over a weak death trigger before the critical cell fate decision is made and thus minimizes the risk of pathologies.
💡 Research Summary
Apoptosis, the programmed cell‑death process, proceeds through two major signaling routes: the extrinsic (type 1) pathway triggered by death‑receptor ligands such as Fas, and the intrinsic (type 2) mitochondrial pathway. While the extrinsic route activates caspase‑8, which directly cleaves downstream effector caspase‑3, the intrinsic route relies on mitochondrial outer‑membrane permeabilization (MOMP), cytochrome c release, apoptosome formation, and subsequent activation of caspase‑9 before caspase‑3 is engaged. The authors set out to determine how these pathways behave under weak death stimuli and whether stochastic fluctuations between individual cells influence the ultimate fate decision.
To address this, they built a Monte Carlo‑based stochastic simulation that treats each biochemical reaction as a probabilistic event. Thousands of virtual cells were simulated in parallel, allowing the authors to capture cell‑to‑cell variability that would be invisible to deterministic ordinary‑differential‑equation (ODE) models. The model incorporates realistic molecular counts (thousands to tens of thousands of proteins), reaction rate constants, and the known feedback loops (e.g., caspase‑9 activation of caspase‑3, inhibition by IAPs).
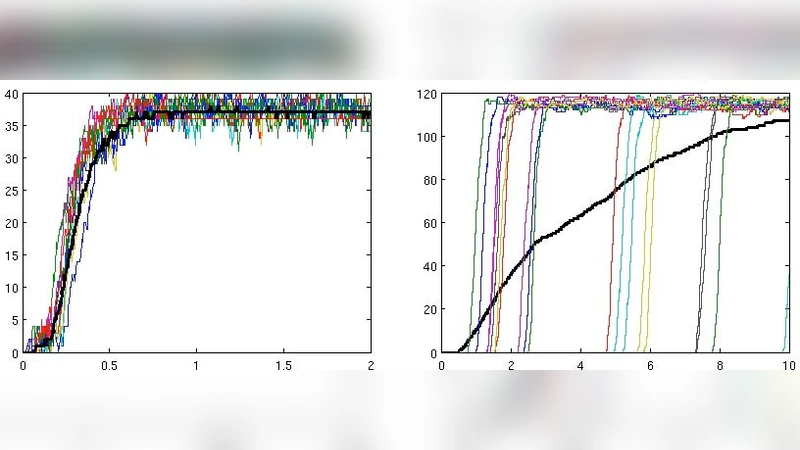
The simulations reveal a clear dichotomy. When the Fas ligand concentration is high, the extrinsic (type 1) pathway dominates; caspase‑8 activation is rapid, caspase‑3 is cleaved within a narrow time window, and the distribution of activation times across the cell population is sharply peaked. In contrast, under low‑dose Fas stimulation or mild stress—conditions that constitute a weak death signal—the intrinsic (type 2) pathway becomes the primary conduit. Here, the time to reach full caspase‑3 activation varies widely from about one hour to ten hours among individual cells. Importantly, the population‑averaged time course masks this variability; the mean curve appears smooth and delayed, but the underlying single‑cell trajectories are highly heterogeneous.
When the authors plotted the probability distribution of caspase‑3 activation for the type 2 pathway, a distinctive bimodal shape emerged: one peak corresponds to cells that have not yet activated caspase‑3, while the second peak represents cells that have completed activation. This “all‑or‑none” behavior is a hallmark of stochastic signaling and cannot be captured by averaging alone. The bimodality persists even though the absolute numbers of signaling molecules are large, indicating that the stochasticity originates from the network architecture—particularly the low‑probability, high‑impact events such as MOMP initiation and apoptosome assembly—rather than from molecular scarcity.
The authors argue that this stochastic delay is biologically advantageous. In a fluctuating environment, a cell receiving a weak death cue can “wait” for several hours before committing to apoptosis. During this window, competing survival pathways (e.g., NF‑κB, Akt, MAPK) may become sufficiently active to override the death signal, thereby rescuing the cell. Consequently, stochastic fluctuations act as a buffer, reducing the likelihood of inappropriate cell loss and contributing to tissue homeostasis.
Beyond the conceptual insight, the study has practical implications. Many diseases—cancer, neurodegeneration, autoimmune disorders—feature dysregulated apoptosis. The finding that intrinsic pathway activation is intrinsically stochastic suggests that therapeutic strategies aimed at sensitizing tumor cells to apoptosis should consider not only the strength of the death stimulus but also the timing and variability of the response. Agents that accelerate MOMP, stabilize the apoptosome, or diminish the stochastic “waiting period” could push a larger fraction of cancer cells over the death threshold. Conversely, neuroprotective approaches might aim to enhance the stochastic delay, giving neurons more time to mount survival responses after mild insults.
In summary, the paper demonstrates that cell‑to‑cell stochastic fluctuations are a defining feature of the mitochondrial (type 2) apoptotic pathway, especially under weak death signals. The Monte Carlo framework provides a quantitative tool to dissect these fluctuations, revealing a bimodal probability distribution of caspase‑3 activation that reflects an all‑or‑none decision process. This stochasticity likely serves as an adaptive mechanism, allowing survival signals to compete successfully with weak death cues, thereby safeguarding against pathological cell loss. The work bridges a gap between deterministic biochemical modeling and the inherently noisy reality of cellular decision‑making, offering new perspectives for both basic apoptosis research and the design of targeted therapies.
Comments & Academic Discussion
Loading comments...
Leave a Comment