Nanomechanical detection of antibiotic-mucopeptide binding in a model for superbug drug resistance
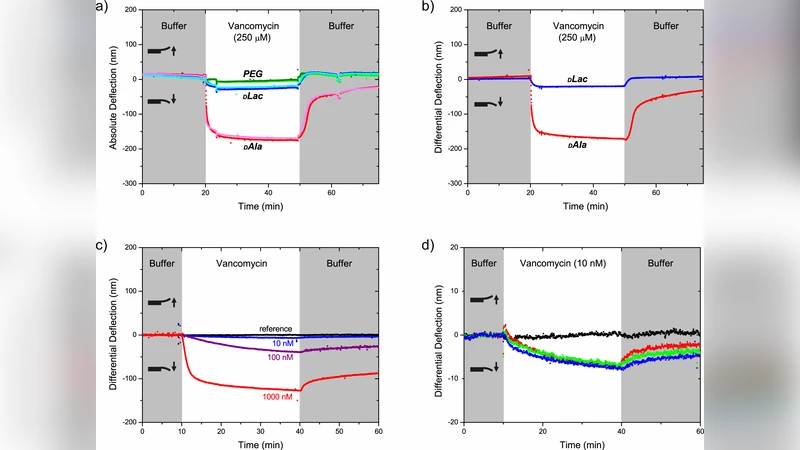
The alarming growth of the antibiotic-resistant superbugs methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococcus (VRE) is driving the development of new technologies to investigate antibiotics and their modes of action. We report the label-free detection of vancomycin binding to bacterial cell wall precursor analogues (mucopeptides) on cantilever arrays, with 10 nM sensitivity and at clinically relevant concentrations in blood serum. Differential measurements quantified binding constants for vancomycin-sensitive and vancomycin-resistant mucopeptide analogues. Moreover, by systematically modifying the mucopeptide density we gain new insights into the origin of surface stress. We propose that stress is a product of a local chemical binding factor and a geometrical factor describing the mechanical connectivity of regions affected by local binding in terms of a percolation process. Our findings place BioMEMS devices in a new class of percolative systems. The percolation concept will underpin the design of devices and coatings to significantly lower the drug detection limit and may also impact on our understanding of antibiotic drug action in bacteria.
💡 Research Summary
The paper presents a label‑free nanomechanical biosensor based on micro‑cantilever arrays that can detect the binding of the glycopeptide antibiotic vancomycin to synthetic analogues of bacterial cell‑wall precursors (mucopeptides) with a sensitivity down to 10 nM. The authors functionalized each cantilever with a self‑assembled monolayer containing either a vancomycin‑sensitive mucopeptide (bearing the canonical D‑Ala‑D‑Ala terminus) or a resistant analogue (bearing a D‑Ala‑D‑Lac substitution). By flowing vancomycin solutions over the devices, they recorded cantilever deflection as a surface‑stress signal, enabling real‑time monitoring of binding events without any fluorescent or radioactive tags.
Key experimental findings include:
-
Ultra‑low detection limit in complex media – The cantilever sensor responded robustly to vancomycin concentrations as low as 10 nM, and the same performance was retained in 10 % human serum, demonstrating that the approach is compatible with clinically relevant sample matrices.
-
Quantitative binding constants – Differential measurements on the two mucopeptide variants yielded dissociation constants (K_D) of ~0.5 µM for the sensitive analogue and ~30 µM for the resistant analogue. These values agree with those obtained by conventional techniques (e.g., surface plasmon resonance) but are obtained here in a single, label‑free assay that directly reports mechanical stress.
-
Systematic density variation – By adjusting the surface density of mucopeptide molecules, the authors discovered a non‑linear relationship between ligand coverage and stress amplitude. Below a critical coverage, the stress signal is negligible; above it, the signal rises sharply, indicating a percolation‑type transition.
-
Percolation model of surface stress – The authors propose that the observed stress is not simply the sum of independent binding events. Instead, it can be expressed as σ_total = σ_b × Φ, where σ_b is the local stress generated by a single binding event and Φ is a geometrical factor describing the mechanical connectivity of the bound region. Φ follows percolation theory: it remains near zero until the ligand density exceeds a threshold (p_c ≈ 0.35 nm⁻² in their system), after which a spanning network of bound molecules transmits stress across the cantilever. This model successfully fits the experimental data and provides a mechanistic explanation for the abrupt amplification of the signal.
-
Implications for device engineering – The percolation framework suggests two routes to improve detection limits: (i) increase the density and uniformity of functional ligands to lower the percolation threshold, and (ii) tailor cantilever mechanical properties (thickness, Young’s modulus) to maximize stress transduction. By combining these strategies, the authors anticipate that sub‑pico‑molar detection could become feasible.
-
Biological relevance – The percolation concept also offers insight into how vancomycin exerts its antibacterial effect. In a native bacterial cell wall, D‑Ala‑D‑Ala termini must be present at sufficient density to form a mechanically connected network that can transmit the binding-induced stress, leading to cell‑wall disruption. The D‑Ala‑D‑Lac resistance mutation effectively breaks this network, preventing percolation and thereby reducing drug efficacy.
Overall, the study demonstrates that nanomechanical cantilever arrays can serve as ultra‑sensitive, label‑free platforms for probing antibiotic–target interactions. By introducing a percolation‑based description of surface stress, the work bridges the gap between molecular binding thermodynamics and macroscopic mechanical readout, opening new avenues for the design of next‑generation BioMEMS diagnostics and for a deeper mechanistic understanding of antibiotic resistance. Future work will likely extend the approach to other drug classes, integrate microfluidic sample handling, and move toward point‑of‑care devices capable of real‑time therapeutic drug monitoring.
Comments & Academic Discussion
Loading comments...
Leave a Comment