Surface instability of sheared soft tissues
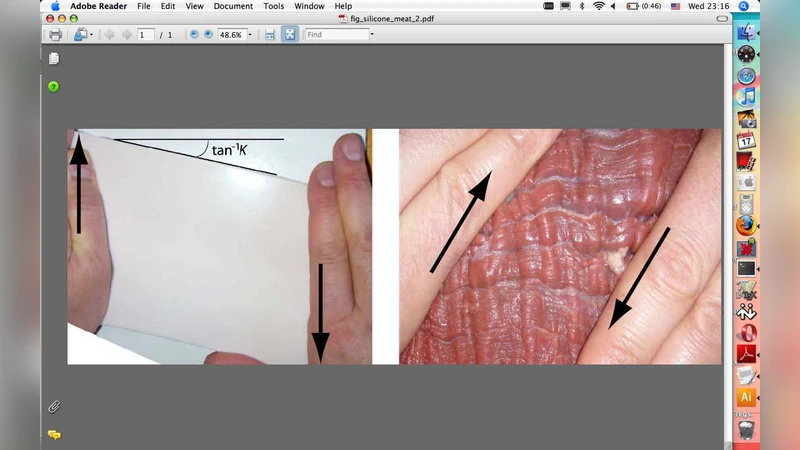
When a block made of an elastomer is subjected to large shear, its surface remains flat. When a block of biological soft tissue is subjected to large shear, it is likely that its surface in the plane of shear will buckle (apparition of wrinkles). One factor that distinguishes soft tissues from rubber-like solids is the presence – sometimes visible to the naked eye – of oriented collagen fibre bundles, which are stiffer than the elastin matrix into which they are embedded but are nonetheless flexible and extensible. Here we show that the simplest model of isotropic nonlinear elasticity, namely the incompressible neo-Hookean model, suffers surface instability in shear only at tremendous amounts of shear, i.e., above 3.09, which corresponds to a 72 degrees angle of shear. Next we incorporate a family of parallel fibres in the model and show that the resulting solid can be either reinforced or strongly weakened with respect to surface instability, depending on the angle between the fibres and the direction of shear, and depending on the ratio E/mu between the stiffness of the fibres and that of the matrix. For this ratio we use values compatible with experimental data on soft tissues. Broadly speaking, we find that the surface becomes rapidly unstable when the shear takes place “against” the fibres, and that as E/mu increases, so does the sector of angles where early instability is expected to occur.
💡 Research Summary
The paper investigates why the surface of a soft biological tissue, unlike that of a rubber‑like elastomer, tends to develop wrinkles when subjected to large shear deformations. The authors first consider the simplest isotropic incompressible hyperelastic model – the neo‑Hookean solid – and perform a linearized incremental analysis of surface waves using the Stroh formalism. By solving the surface‑instability (bifurcation) condition they find that the neo‑Hookean block remains stable up to a very large shear amount γ≈3.09, corresponding to a shear angle of about 72°. This result is far beyond the shear levels at which wrinkles are observed in real tissues, indicating that isotropy alone cannot explain the phenomenon.
To capture the distinctive microstructure of soft tissues, the authors enrich the model with a family of parallel collagen fibres embedded in the elastin matrix. The matrix still follows the incompressible neo‑Hookean law (shear modulus μ), while the fibres are modeled as linearly elastic with modulus E. The key dimensionless parameter is the stiffness ratio E/μ, which the authors choose within the range reported for biological tissues (roughly 5–30). The fibre direction is denoted by an angle α with respect to a fixed reference, and the shear direction by β; the relative angle Δ=β−α governs the interaction between the imposed shear and the fibre reinforcement.
Using the same incremental framework, the total strain‑energy density W_total = μ(I₁−3)/2 + (E/2)(n·C·n−1)² (where n is the unit fibre vector and C the right Cauchy‑Green tensor) is linearized about a finite simple‑shear pre‑deformation. The resulting eigenvalue problem yields a characteristic equation whose roots λ determine the decay of perturbations away from the free surface. Surface instability occurs when a root crosses the real axis, i.e., when the real part of λ becomes zero. By solving this condition numerically for various Δ and E/μ, the authors map the critical shear γ_c(Δ, E/μ).
The analysis reveals two contrasting regimes:
-
Reinforcement regime (Δ≈0) – when the fibres are aligned with the shear direction, they bear most of the shear load. The critical shear γ_c increases dramatically compared with the pure neo‑Hookean case, indicating a highly stable surface. For E/μ≈10 the instability threshold can exceed γ≈5.5 (≈80°).
-
Weakening regime (Δ≈±90°) – when the shear acts “against” the fibres, the fibres are stretched rather than sheared, producing large normal stresses that concentrate at the surface. In this configuration γ_c drops sharply; even modest shears of 30°–45° (γ≈0.6–1.0) can trigger surface wrinkling. The weakening effect intensifies as E/μ grows, expanding the angular sector Δ over which early instability occurs.
Thus, the fibre‑matrix stiffness ratio controls not only the magnitude of the critical shear but also the angular width of the unstable region. The authors compare these theoretical predictions with experimental observations on skin, arterial walls, and tendon, noting that the measured E/μ values and typical fibre orientations fall within the regimes where early surface buckling is expected.
From a biomechanical perspective, the findings have several implications. Surgical procedures that impose shear on soft tissues (e.g., flap rotation, tendon grafting) should account for the underlying fibre architecture; cutting across fibres can dramatically lower the shear threshold for surface damage. In tissue engineering, designing scaffolds with tailored fibre orientation and stiffness can either suppress unwanted wrinkling (by aligning fibres with anticipated shear) or promote controlled surface patterning (by orienting fibres orthogonal to shear). Moreover, non‑invasive imaging of surface topography could serve as a diagnostic proxy for fibre alignment and tissue health.
In summary, the paper demonstrates that the neo‑Hookean model alone predicts surface stability only at unrealistically high shear, whereas incorporating a simple anisotropic fibre reinforcement reproduces the early onset of wrinkling observed in real soft tissues. The critical shear depends sensitively on the angle between the shear plane and the fibre direction and on the stiffness ratio E/μ. This work provides a clear, quantitative framework for understanding shear‑induced surface instability in biological soft tissues and offers practical guidance for clinical handling and biomaterial design.
Comments & Academic Discussion
Loading comments...
Leave a Comment