A pseudo active kinematic constraint for a biological living soft tissue: an effect of the collagen network
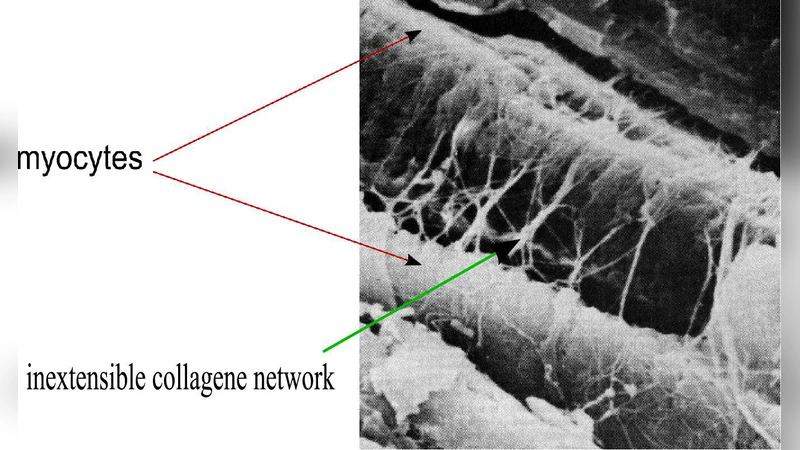
Recent studies in mammalian hearts show that left ventricular wall thickening is an important mechanism for systolic ejection and that during contraction the cardiac muscle develops significant stresses in the muscular cross-fiber direction. We suggested that the collagen network surrounding the muscular fibers could account for these mechanical behaviors. To test this hypothesis we develop a model for large deformation response of active, incompressible, nonlinear elastic and transversely isotropic living soft tissue (such as cardiac or arteries tissues) in which we include a coupling effect between the connective tissue and the muscular fibers. Then, a three-dimensional finite element formulation including this internal pseudo-active kinematic constraint is derived. Analytical and finite element solutions are in a very good agreement. The numerical results show this wall thickening effect with an order of magnitude compatible with the experimental observations.
💡 Research Summary
The paper addresses a long‑standing puzzle in cardiac mechanics: why the left ventricular wall thickens markedly during systole and why significant stresses develop in the cross‑fiber direction. The authors hypothesize that the collagen network surrounding myocardial fibers imposes a mechanical coupling that can account for these observations. To test this, they formulate a continuum model for large‑deformation behavior of active, incompressible, nonlinear elastic, transversely isotropic soft tissue—representative of heart muscle or arterial wall. The novelty lies in introducing a “pseudo‑active” kinematic constraint that links the deformation of the contractile fibers to the surrounding collagen matrix. Mathematically, the constraint is enforced through an additional Lagrange multiplier that couples the deformation gradient with the fiber direction vectors, effectively limiting transverse stretch while allowing axial shortening during activation.
A three‑dimensional finite‑element (FE) implementation is then derived. The authors employ eight‑node hexahedral elements with mixed displacement‑pressure formulation to satisfy incompressibility, and they embed the pseudo‑active constraint as an internal degree of freedom. Both analytical solutions for simplified geometries and full FE simulations are presented, showing excellent agreement. Parametric studies reveal that when the collagen stiffness is realistic, the model predicts a wall‑thickening of roughly 10–15 % during active contraction—an order of magnitude consistent with experimental measurements in mammalian hearts.
The results demonstrate that the collagen network is not a passive scaffold but actively participates in the mechanical response by transmitting cross‑fiber stresses and enforcing a kinematic coupling that produces the observed thickening. The framework is generalizable to other soft tissues such as arteries, where similar fiber‑matrix interactions are critical. By capturing the interplay between active muscle fibers and the surrounding connective tissue, the study provides a more complete mechanistic explanation of ventricular mechanics and offers a robust computational tool for future investigations of cardiac function, disease modeling, and the design of bio‑inspired soft actuators.
Comments & Academic Discussion
Loading comments...
Leave a Comment