Evolutionary game theory elucidates the role of glycolysis in glioma progression and invasion
Tumour progression has been described as a sequence of traits or phenotypes that cells have to acquire if the neoplasm is to become an invasive and malignant cancer. Although the genetic mutations that lead to these phenotypes are random, the process by which some of these mutations become successful and spread is influenced by the tumour microenvironment and the presence of other phenotypes. It is thus likely that some phenotypes that are essential in tumour progression will emerge in the tumour population only with the prior presence of other different phenotypes. In this paper we use evolutionary game theory to analyse the interactions between three different tumour cell phenotypes defined by autonomous growth, anaerobic glycolysis, and cancer cell invasion. The model allows to understand certain specific aspects of glioma progression such as the emergence of diffuse tumour cell invasion in low-grade tumours. We find that the invasive phenotype is more likely to evolve after the appearance of the glycolytic phenotype which would explain the ubiquitous presence of invasive growth in malignant tumours. The result suggests that therapies which increase the fitness cost of switching to anaerobic glycolysis might decrease the probability of the emergence of more invasive phenotypes
💡 Research Summary
In this paper the authors apply evolutionary game theory (EGT) to investigate how three phenotypic strategies commonly observed in glioma—autonomous growth (AG), anaerobic glycolysis (GLY), and invasion/motility (INV)—interact and shape tumor progression. The model assumes that all tumor cells initially possess the AG phenotype, which confers independence from external growth factors. Mutations may give rise to either a glycolytic phenotype, which trades off energetic efficiency for survival under hypoxia and acidifies the micro‑environment, or an invasive phenotype, which incurs a motility cost but allows cells to colonize new space.
A 3 × 3 payoff matrix quantifies the fitness change experienced by a focal cell when interacting with another cell of a given phenotype. Three parameters control the matrix:
- k – the fitness cost of adopting the less efficient glycolytic metabolism. Larger k makes GLY less advantageous.
- n – a dual term that represents the loss of fitness for a non‑glycolytic cell exposed to acidity and the gain for a glycolytic cell that acidifies the environment. Higher n intensifies the advantage of GLY and the disadvantage of AG in acidic neighborhoods.
- c – the cost of motility for INV cells, reflecting reduced proliferation while moving. Higher c makes invasion less favorable.
Two analytical scenarios are explored.
Scenario 1 (AG vs. INV). Using replicator dynamics the authors find that if the motility cost satisfies c ≥ 0.5, the AG population is immune to invasion because the payoff of an AG‑AG interaction (½) exceeds the payoff of an INV‑AG interaction (1 − c). When c is lower, a polymorphic equilibrium can arise where both phenotypes coexist with equal fitness. This result highlights that the physical constraints of the brain (white‑matter tracts reducing c) can facilitate invasion even in the absence of glycolysis.
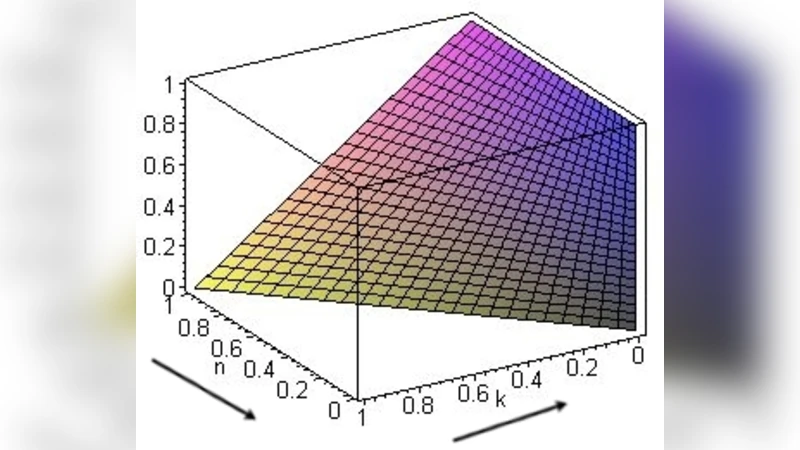
Scenario 2 (AG, GLY, INV together). The authors solve for an interior equilibrium where the average fitness of all three strategies is equal. The equilibrium frequencies depend on k, n, and c. Crucially, the proportion of invasive cells (p) increases when k is low (glycolysis cheap) and n is high (acidic environment strongly penalizes non‑glycolytic cells). In this regime, the presence of GLY cells effectively lowers the relative cost of motility, allowing INV cells to dominate. Conversely, raising k or lowering n reduces p and favors AG or GLY cells.
Figures illustrate these dependencies: Figure 1 shows p as a function of k and n, revealing a region of low k/high n where INV cells displace the others. Figure 2 presents the AG fraction for four values of c (0, 0.25, 0.5, 0.75). As c rises, the AG fraction shrinks and GLY cells become more prevalent, indicating that high motility costs push the tumor toward a glycolytic, less invasive state.
The discussion places the mathematical findings in a biological context. Gliomas are uniquely invasive even at low grade, which the model attributes to the brain’s low motility cost (c) and the early appearance of glycolytic cells that acidify the micro‑environment. The authors note that in many epithelial cancers, higher physical barriers raise c, making invasion less common without a glycolytic pre‑condition. Clinical imaging (FDG‑PET, MRS) supports the link between glycolysis and malignancy grade in glioma, aligning with the model’s prediction that glycolytic (malignant) tumors will almost inevitably acquire an invasive phenotype.
Therapeutically, the model suggests that increasing the cost of glycolysis (raising k) – for example by improving tissue oxygenation or using glycolysis inhibitors – could reduce the likelihood of invasion and subsequent metastasis. Conversely, interventions that inadvertently lower motility cost (e.g., matrix‑degrading agents) might promote invasion. The authors acknowledge limitations, notably the absence of explicit spatial structure, and propose future extensions incorporating dynamic cost functions and additional phenotypes defined by molecular alterations.
In summary, the paper demonstrates that a simple EGT framework can capture key aspects of glioma evolution: glycolysis tends to precede and facilitate invasion, and the balance of metabolic and motility costs determines the stable composition of tumor cell phenotypes. These insights provide a theoretical basis for metabolic‑targeted therapies aimed at curbing tumor invasiveness.
Comments & Academic Discussion
Loading comments...
Leave a Comment