A Mathematical Model of Chaotic Attractor in Tumor Growth and Decay
We propose a strange-attractor model of tumor growth and metastasis. It is a 4-dimensional spatio-temporal cancer model with strong nonlinear couplings. Even the same type of tumor is different in every patient both in size and appearance, as well as in temporal behavior. This is clearly a characteristic of dynamical systems sensitive to initial conditions. The new chaotic model of tumor growth and decay is biologically motivated. It has been developed as a live Mathematica demonstration, see Wolfram Demonstrator site: http://demonstrations.wolfram.com/ChaoticAttractorInTumorGrowth/ Key words: Reaction-diffusion tumor growth model, chaotic attractor, sensitive dependence on initial tumor characteristics
💡 Research Summary
The paper proposes a novel chaotic attractor model for tumor growth and metastasis, extending a previously established four‑dimensional reaction‑diffusion framework. The authors begin by reviewing the multiscale Anderson‑Chaplain model, which couples tumor cell density (n), matrix‑metalloprotein concentration (f), matrix‑degradative enzyme concentration (m), and oxygen concentration (c) through diffusion, haptotaxis, production, and decay terms. After non‑dimensionalisation, the system is expressed as equations (5)–(8) with parameters calibrated from literature values.
When the spatial derivatives are omitted, the resulting ordinary differential equations (11)–(14) exhibit essentially linear, uncoupled dynamics: n remains constant, and the remaining variables evolve independently, failing to capture the observed heterogeneity among patients. To introduce genuine nonlinear interaction, the authors add four biologically motivated parameters—α, β, γ, and δ—representing, respectively, the proliferative versus non‑proliferative cell volume fraction, glucose level, total cell number, and surface diffusion saturation. The modified temporal system (15)–(18) replaces the simple decay of f with a term αη(m − f), couples m and c through a bilinear ν f m term, and incorporates γ into the m‑equation, thereby creating feedback loops reminiscent of the Lorenz equations.
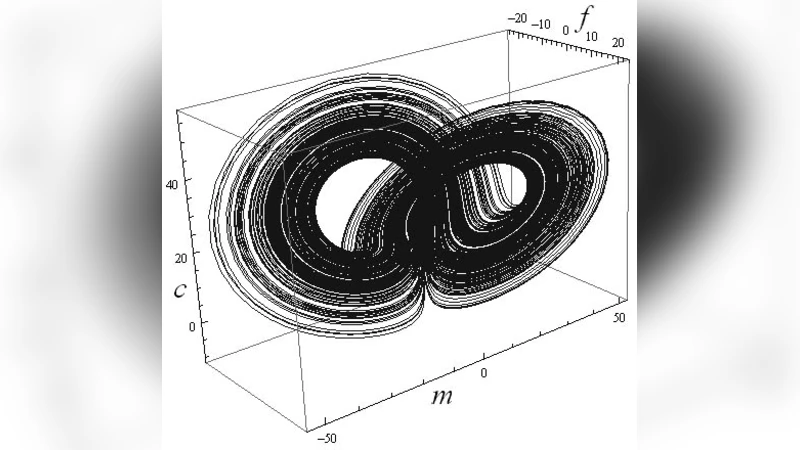
Numerical integration with α = 0.06, β = 0.05, γ = 26.5, and δ = 40 produces a three‑dimensional strange attractor in the (f, m, c) subspace, visually analogous to the classic Lorenz butterfly (Figure 1). This demonstrates that the tumor system can exhibit sensitive dependence on initial conditions, long‑term unpredictability, and fractal‑like dynamics—features that align with clinical observations of patient‑specific tumor growth patterns and treatment responses.
The authors then reincorporate the nonlinear terms into the original partial differential equations, yielding a spatio‑temporal model (19)–(22) that retains the chaotic “butterfly” behavior while preserving diffusion and haptotaxis. They argue that this multiscale chaotic model offers a more realistic representation of tumor invasion, accounting for both stochastic‑like long‑term variability and the influence of microenvironmental factors.
Biologically, the parameters α, β, γ, and δ are linked to measurable quantities: α to the ratio of proliferating to quiescent cells, β to nutrient availability, γ to cell density (which correlates with Cdk inhibitor p27 levels), and δ to extracellular matrix remodeling. The paper suggests that therapeutic strategies could target these parameters—e.g., re‑programming cancer stem cells, activating p27, or modulating SATB1 expression—to either suppress chaos (using OGY‑type control) or deliberately induce it (anti‑control) to destabilize malignant dynamics.
In conclusion, the study presents a plausible chaotic multiscale tumor‑invasion model, demonstrating that modest nonlinear couplings can generate a strange attractor and thereby explain inter‑patient variability and the difficulty of predicting tumor evolution. While the approach is conceptually appealing, it currently lacks experimental validation of the new parameters, omits spatial heterogeneity in the chaotic analysis, and does not address how robust the chaotic regime is under realistic biological noise. Future work should focus on calibrating α, β, γ, and δ with patient data, extending the model to fully coupled PDE‑ODE hybrids, and testing chaos‑control algorithms in silico and, eventually, in pre‑clinical settings.
Comments & Academic Discussion
Loading comments...
Leave a Comment