Potential Phytoextraction with in-vitro regenerated plantlets of Brassica juncea (L.) Czern. in presence of CdCl$_2$: Cadmium accumulation and physiological parameter measurement
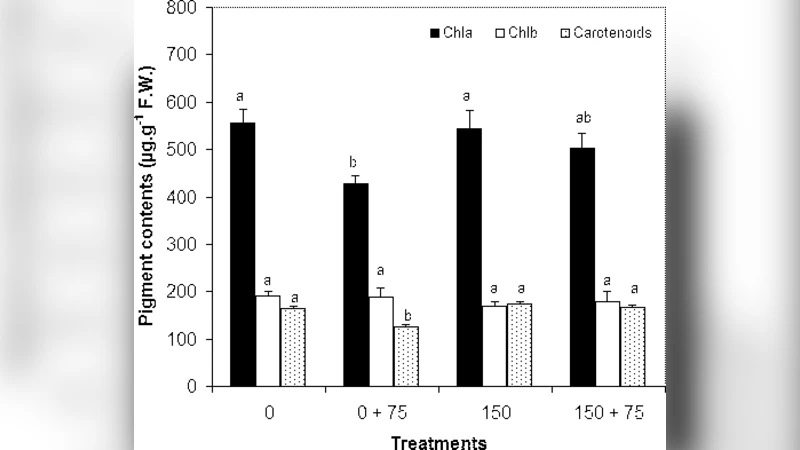
Heavy metal contamination of agricultural land is partly responsible for limiting crop productivity. Cd$^{2+}$ is known as a non-essentiel HM that can be harmful to plants even at low concentrations. Brassica juncea (L.) is able to accumulate more than 400 $\mu$g.g$^{-1}$ D.W. in the shoot, a physiological trait which may be exploited for the phytoremediation of contaminated soils and waters. . The application of 75 $\mu$M CdCl$_2$ for three days does not show any effect in the B. juncea growth parameters (F.W. and D.W.) whatever the type of plantlets. This application decreases also the contents of chlorophyll a, carotenoids and Chl a/b ratio (2.26) for plantlets regenerated in the absence of CdCl$_2$ but not those of plantlets regenerated in its presence. Roots have the highest contents (3071; 1544 $\mu$g.g$^{-1}$ D.W.) followed by stems (850; 687$\mu$g.g$^{-1}$ D.W.) and leaves (463; 264$\mu$g.g$^{-1}$ D.W.) respectively. In our conditions, we suggest that the low accumulation in the plantlets regenerated in the presence of CdCl$_2$ by the means of in-vitro regeneration technology is still benefical, to some extent, for the phytoextraction process and seems to be an interesting technology that allows the cultivation of these plantlets in contaminated soils with low accumulation of metal in their shoots and probably in their seeds used in many food technologies.
💡 Research Summary
The study investigates the potential of using in‑vitro regenerated Brassica juncea (L.) plantlets for cadmium (Cd) phytoextraction and assesses how prior exposure to Cd during tissue culture influences subsequent Cd stress responses. Two groups of plantlets were produced: one regenerated on a standard Murashige‑Skoog medium without Cd (referred to as “non‑exposed”) and another regenerated on the same medium supplemented with 75 µM CdCl₂ (referred to as “Cd‑pre‑exposed”). After regeneration, both groups were subjected to a uniform Cd challenge—75 µM CdCl₂ for three days—in hydroponic culture. The experimental design allowed a direct comparison of growth parameters, photosynthetic pigment content, and tissue‑specific Cd accumulation between plantlets that had never encountered Cd and those that had been conditioned by Cd during their early development.
Growth measurements (fresh weight and dry weight) showed no statistically significant differences between the two groups after the three‑day Cd exposure, indicating that B. juncea possesses a relatively high tolerance to this Cd concentration over short periods. However, pigment analysis revealed a marked divergence. In the non‑exposed plantlets, Cd treatment caused a pronounced decline in chlorophyll a and carotenoid concentrations, and the chlorophyll a/b ratio dropped to 2.26, reflecting damage to the photosynthetic apparatus, likely through inhibition of chlorophyll biosynthetic enzymes and enhanced oxidative stress. By contrast, Cd‑pre‑exposed plantlets maintained near‑baseline pigment levels, suggesting that prior Cd exposure induced protective mechanisms—such as up‑regulation of metal‑binding proteins (e.g., phytochelatins), antioxidant enzymes, and stress‑responsive transcription factors—that mitigated the acute toxic effects of subsequent Cd exposure.
Elemental analysis by ICP‑MS demonstrated a clear tissue hierarchy for Cd accumulation: roots > stems > leaves. In non‑exposed plantlets, Cd concentrations reached 3071 µg g⁻¹ dry weight (DW) in roots, 850 µg g⁻¹ DW in stems, and 463 µg g⁻¹ DW in leaves. Cd‑pre‑exposed plantlets accumulated roughly half of these amounts (1544 µg g⁻¹ DW in roots, 687 µg g⁻¹ DW in stems, 264 µg g⁻¹ DW in leaves). This reduction indicates that early Cd exposure can modulate metal translocation pathways, possibly by enhancing sequestration in root cell walls, increasing vacuolar compartmentalization, or altering expression of metal transporter genes (e.g., HMA, NRAMP families). The lower leaf Cd load is particularly relevant for food safety, as it suggests that shoots harvested from such plantlets would contain Cd levels below critical thresholds for human consumption.
The authors discuss the implications of these findings for phytoremediation strategies. Traditional phytoextraction relies on high‑accumulating genotypes that concentrate metals in harvestable above‑ground biomass, followed by safe disposal or processing of the contaminated biomass. In contrast, the present approach aims to generate plantlets that tolerate Cd‑contaminated soils while limiting Cd translocation to edible tissues. This “low‑accumulation” phenotype could enable the cultivation of B. juncea on marginal, Cd‑polluted lands without compromising crop safety, thereby providing both ecological remediation and economic utility. Moreover, the study highlights the utility of tissue‑culture‑based pre‑conditioning as a tool to reprogram plant metal homeostasis.
Limitations include the short exposure duration (three days) and the hydroponic setting, which does not fully replicate the complex physicochemical interactions present in field soils (e.g., pH, organic matter binding, microbial activity). Consequently, long‑term field trials are necessary to validate the durability of the induced tolerance and to quantify actual Cd removal rates from contaminated soils.
In conclusion, the research demonstrates that in‑vitro regeneration of B. juncea in the presence of Cd can produce plantlets that retain normal growth, preserve photosynthetic pigment integrity, and exhibit reduced Cd translocation to shoots. This strategy offers a promising avenue for developing crops capable of thriving on Cd‑contaminated sites while minimizing food‑chain contamination, thereby contributing to sustainable phytoremediation and agricultural practices.
Comments & Academic Discussion
Loading comments...
Leave a Comment