Epidermal corneocytes: dead guards of the hidden treasure
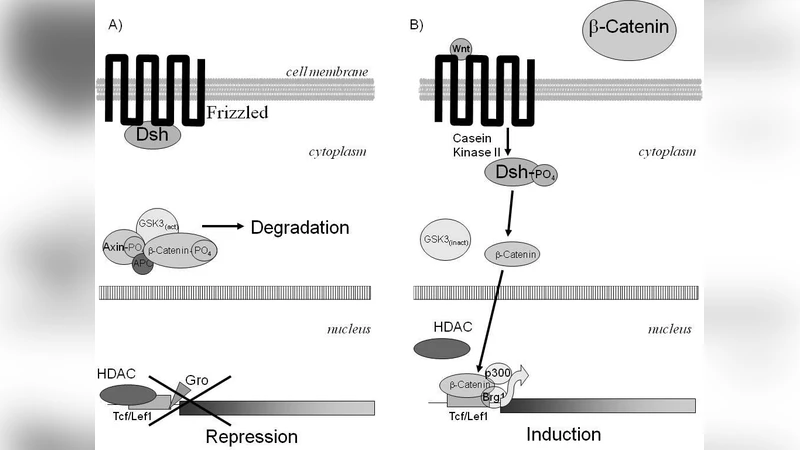
Gradual transformation of the epidermal stem cells to corneocytes involves a chain of chronologically well-arranged events that mostly stimulated locally by their neighbors. Cell diversity that observed during the differentiation through the different epidermal cell layers included the consisted changes of cell shape, intercellular contacts and proliferation. However, the most dramatically these changes appeared at the molecular level through gene expression, catalysis and intraprotein interactions. The proposed review explains these changes by switching systemic transcription factors that unlike their counterparts those role is limited to a contribution to gene expression also prepare cells to the next step of differentiation via modification of the chromatin pattern . Since primary epidermal keratinocytes are one of the most easy available type of the stem cells, a better understanding of the epidermal differentiation will benefit the research in the other areas by a discovery of basic coordinating mechanisms that stand behind such distinct molecular events as cell signaling and gene expression, and formulate basic principles for a smart therapeutic correction of the metabolism.
💡 Research Summary
The paper provides a comprehensive review of epidermal differentiation, focusing on how epidermal stem cells gradually transform into terminally dead corneocytes that constitute the skin’s protective barrier. It begins by outlining the hierarchical organization of the epidermis into basal, spinous, granular, and cornified layers, each characterized by distinct morphological and molecular signatures. The authors emphasize that the differentiation cascade is orchestrated by a tightly regulated sequence of transcriptional, epigenetic, structural, and metabolic events, most of which are driven by local cell‑cell communication.
At the transcriptional level, the review highlights master regulators such as p63, KLF4, and GRHL3 that maintain basal cell proliferation and initiate early differentiation. As cells ascend, p63 expression wanes while AP‑1 complexes take over, activating genes encoding involucrin, loricrin, and filaggrin—key components of the cornified envelope. The authors argue that this “transcription factor switch” is not merely a change in gene activation but also a trigger for chromatin remodeling. They describe how the loss of p63 facilitates recruitment of Polycomb repressive complex 2 (PRC2) and deposition of H3K27me3 marks, leading to stable silencing of proliferative genes. Concurrently, SWI/SNF chromatin remodelers are displaced, and DNA methylation patterns are reshaped by DNMTs and TET enzymes, cementing the differentiated state.
Structural remodeling accompanies the transcriptional shift. Early epidermal layers rely on strong desmosomal adhesion (Desmoglein‑1/2) and E‑cadherin–mediated contacts, whereas the suprabasal layers up‑regulate tight‑junction proteins such as claudin‑1, claudin‑4, and occludin to reinforce barrier integrity. Cytoskeletal dynamics also change: actin‑myosin contractility diminishes, and keratin 1/10 filaments become predominant, flattening the cells and preparing them for dehydration.
Metabolically, the review points out a decisive switch from glycolysis‑driven biosynthesis in proliferative keratinocytes to an AMPK‑activated, mTORC1‑suppressed state in differentiating cells. This shift favors fatty‑acid β‑oxidation and the accumulation of natural moisturizing factors derived from filaggrin breakdown, which are essential for maintaining hydration in the outermost cornified layer.
The authors propose an integrated model in which transcription factor turnover, epigenetic reprogramming, adhesion/cytoskeletal reorganization, and metabolic rewiring act synergistically to produce the “dead guards” of the skin. They argue that understanding this coordinated program has broad implications: it can illuminate the pathogenesis of common skin disorders (psoriasis, atopic dermatitis, lupus), guide the development of targeted therapeutics (e.g., p63 agonists, PRC2 inhibitors), and improve tissue‑engineered skin substitutes by providing precise cues for in‑vitro corneocyte formation.
In conclusion, the paper positions epidermal keratinocytes as an accessible model for stem‑cell biology and underscores that dissecting the molecular choreography of epidermal differentiation will yield fundamental principles applicable to regenerative medicine, disease modeling, and smart metabolic correction strategies.
Comments & Academic Discussion
Loading comments...
Leave a Comment