Chaperones as integrators of cellular networks: Changes of cellular integrity in stress and diseases
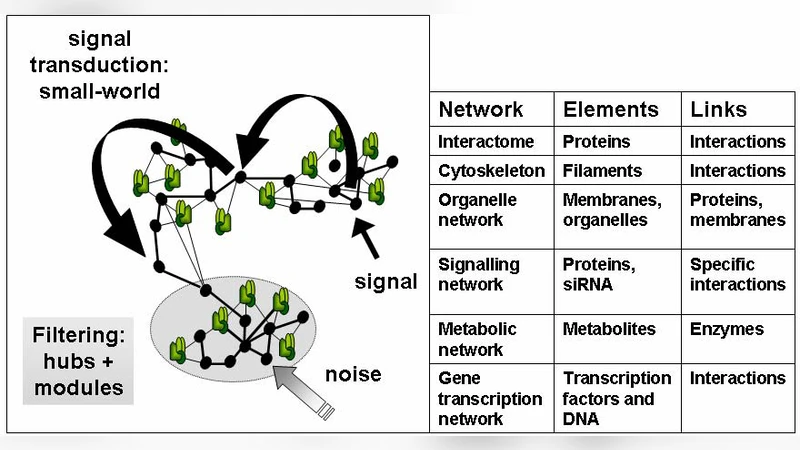
Cellular networks undergo rearrangements during stress and diseases. In un-stressed state the yeast protein-protein interaction network (interactome) is highly compact, and the centrally organized modules have a large overlap. During stress several original modules became more separated, and a number of novel modules also appear. A few basic functions, such as the proteasome preserve their central position. However, several functions with high energy demand, such the cell-cycle regulation loose their original centrality during stress. A number of key stress-dependent protein complexes, such as the disaggregation-specific chaperone, Hsp104, gain centrality in the stressed yeast interactome. Molecular chaperones, heat shock, or stress proteins form complex interaction networks (the chaperome) with each other and their partners. Here we show that the human chaperome recovers the segregation of protein synthesis-coupled and stress-related chaperones observed in yeast recently. Examination of yeast and human interactomes shows that (1) chaperones are inter-modular integrators of protein-protein interaction networks, which (2) often bridge hubs and (3) are favorite candidates for extensive phosphorylation. Moreover, chaperones (4) become more central in the organization of the isolated modules of the stressed yeast protein-protein interaction network, which highlights their importance in the de-coupling and re-coupling of network modules during and after stress. Chaperone-mediated evolvability of cellular networks may play a key role in cellular adaptation during stress and various polygenic and chronic diseases, such as cancer, diabetes or neurodegeneration.
💡 Research Summary
The paper investigates how cellular protein‑protein interaction (PPI) networks are reorganized under stress and disease conditions, focusing on molecular chaperones as network integrators. Using the yeast Saccharomyces cerevisiae interactome as a model, the authors first compare the topology of the network in an unstressed state with that after exposure to heat shock, oxidative stress, or other perturbations. In the basal condition the interactome is highly compact: modules overlap extensively, the average shortest path is short, and a few core hubs (e.g., proteasome components) dominate centrality. When stress is applied, the network undergoes a pronounced modularization. Original modules become more isolated, new stress‑specific modules appear, and the overall clustering coefficient rises while hub centrality shifts. Functions that demand high energy, such as cell‑cycle regulation, lose their central positions, whereas low‑energy maintenance processes (e.g., proteasomal degradation) retain hub status.
A central finding is that chaperones act as inter‑modular bridges. The authors map chaperone–protein interactions and demonstrate that chaperones frequently connect distinct hubs, effectively stitching together otherwise separate modules. This bridging role is reinforced by a systematic analysis of phosphorylation data: chaperones are among the most heavily phosphorylated proteins, indicating that they are prime targets for rapid post‑translational regulation. Phosphorylation can thus modulate chaperone activity, allowing swift re‑wiring of signaling and stress‑response pathways.
The study extends the analysis to the human chaperome. Comparative network reconstruction shows that, like yeast, human chaperones segregate into two major groups: synthesis‑coupled chaperones (e.g., Hsp70/Hsp90 complexes) and stress‑responsive chaperones (e.g., small Hsps such as Hsp27 and αB‑crystallin). This dual compartmentalization is evolutionarily conserved and suggests a universal strategy for rapid network adaptation. Moreover, disease‑associated mutations frequently map onto chaperone nodes, underscoring their relevance in pathological network remodeling.
Finally, the authors propose a “decoupling‑recoupling” model. During stress, chaperones become more central within isolated modules, facilitating the disassembly of damaged complexes and the assembly of protective ones (e.g., disaggregation complexes containing Hsp104). After stress removal, chaperones help re‑couple modules, restoring the compact, efficient network architecture. This dynamic role positions chaperones as key drivers of cellular evolvability, enabling cells to explore alternative network configurations without compromising viability.
Implications for disease are profound. In cancer, where signaling networks are rewired, chaperone‑mediated hub bridging may sustain oncogenic pathways. In metabolic disorders such as diabetes, altered chaperone activity could affect the modularity of insulin‑signaling networks. In neurodegenerative diseases, the failure of chaperone‑driven decoupling may exacerbate protein aggregation and network collapse. Consequently, targeting chaperones—or their phosphorylation regulators—offers a network‑centric therapeutic avenue that could modulate multiple disease‑related pathways simultaneously.
In summary, the paper provides a comprehensive systems‑biology perspective on how molecular chaperones integrate, reorganize, and stabilize cellular interaction networks under stress and disease, highlighting their potential as strategic nodes for future therapeutic interventions.
Comments & Academic Discussion
Loading comments...
Leave a Comment