Determinants of bistability in induction of the Escherichia coli lac operon
We have developed a mathematical model of regulation of expression of the Escherichia coli lac operon, and have investigated bistability in its steady-state induction behavior in the absence of external glucose. Numerical analysis of equations describing regulation by artificial inducers revealed two natural bistability parameters that can be used to control the range of inducer concentrations over which the model exhibits bistability. By tuning these bistability parameters, we found a family of biophysically reasonable systems that are consistent with an experimentally determined bistable region for induction by thio-methylgalactoside (Ozbudak et al. Nature 427:737, 2004). The model predicts that bistability can be abolished when passive transport or permease export becomes sufficiently large; the former case is especially relevant to induction by isopropyl-beta, D-thiogalactopyranoside. To model regulation by lactose, we developed similar equations in which allolactose, a metabolic intermediate in lactose metabolism and a natural inducer of lac, is the inducer. For biophysically reasonable parameter values, these equations yield no bistability in response to induction by lactose; however, systems with an unphysically small permease-dependent export effect can exhibit small amounts of bistability for limited ranges of parameter values. These results cast doubt on the relevance of bistability in the lac operon within the natural context of E. coli, and help shed light on the controversy among existing theoretical studies that address this issue. The results also suggest an experimental approach to address the relevance of bistability in the lac operon within the natural context of E. coli.
💡 Research Summary
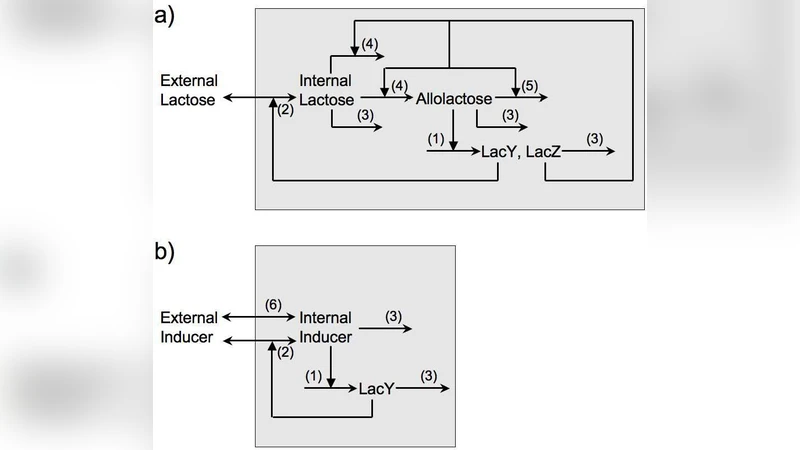
The authors present a comprehensive mathematical analysis of the regulatory network governing the Escherichia coli lac operon, with a focus on whether the system can exhibit bistability (the coexistence of two stable steady‑states) under physiologically relevant conditions. The study is divided into two complementary modeling efforts. First, they construct a set of ordinary differential equations describing the dynamics of lac transcription when the cell is exposed to artificial inducers such as thio‑methyl‑β‑D‑galactoside (TMG) or isopropyl‑β‑D‑thiogalactopyranoside (IPTG). The model incorporates three essential processes: (i) repression by the LacI repressor, (ii) permease‑mediated uptake of the inducer, and (iii) permease‑mediated export (or “reverse transport”) of the inducer. By numerically solving the steady‑state equations across a wide range of external inducer concentrations, the authors identify two dimensionless “bistability parameters.” The first parameter quantifies the relative strength of permease‑dependent uptake versus passive diffusion (or leakage) out of the cell; the second captures the efficiency of permease‑mediated export. Systematic variation of these parameters shows that the width and location of the bistable region in the inducer‑response curve can be precisely tuned. In particular, when export is strong enough, the S‑shaped response collapses into a single monotonic curve, eliminating bistability. Conversely, when export is negligible and uptake dominates, a broad bistable interval emerges. The authors demonstrate that by adjusting these two parameters they can reproduce the experimentally observed bistable window for TMG reported by Ozbudak et al. (Nature 427:737, 2004). They also predict that IPTG, which is not metabolized and has very low export, should retain bistability unless passive diffusion becomes unusually large (e.g., at high temperature or in mutants with increased membrane permeability).
The second part of the paper extends the framework to the natural inducer, lactose. Here the model is expanded to include lactose hydrolysis by β‑galactosidase (LacZ) into allolactose, the true intracellular inducer, and the subsequent binding of allolactose to LacI. The equations also contain permease‑mediated lactose uptake and a term for permease‑dependent export of allolactose. Using parameter values drawn from the literature (Km and Vmax for LacY, LacZ catalytic rates, allolactose‑LacI dissociation constant, etc.), the authors perform a steady‑state bifurcation analysis. Across the biologically plausible parameter space, the lactose‑driven system displays a single stable steady‑state for any external lactose concentration; the characteristic hysteresis associated with bistability never appears. Only when the export term is artificially reduced to unrealistically low values does a narrow bistable region emerge, and even then the range of inducer concentrations supporting bistability is extremely limited. This suggests that, under normal physiological conditions, the lac operon does not exploit bistability when induced by its natural substrate.
The paper therefore reaches two major conclusions. First, artificial inducers can generate bistability, and the extent of this behavior is governed primarily by the balance between permease‑mediated uptake, passive diffusion, and permease‑mediated export. Second, for the natural inducer lactose, realistic kinetic parameters do not support bistability, casting doubt on earlier theoretical claims that the lac system functions as a bistable switch in its native context. To test these predictions experimentally, the authors propose manipulating permease expression levels, creating mutants with diminished export activity, or altering membrane permeability, and then measuring single‑cell fluorescence distributions of a lac‑promoter reporter. Such experiments would directly assess whether the lac operon can occupy two distinct expression states in a homogeneous population when induced by lactose.
Overall, the study provides a clear mechanistic explanation for why bistability is observed with synthetic inducers but is unlikely to be biologically relevant with lactose. It reconciles conflicting theoretical reports, offers concrete experimental strategies, and deepens our understanding of how feedback, transport, and metabolic conversion shape the dynamical landscape of gene regulation in bacteria.
Comments & Academic Discussion
Loading comments...
Leave a Comment