Point Mutations Effects on Charge Transport Properties of the Tumor-Suppressor Gene p53
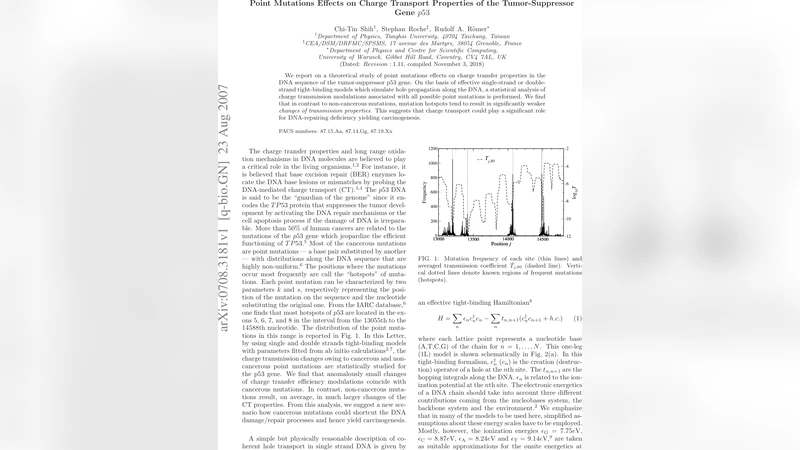
We report on a theoretical study of point mutations effects on charge transfer properties in the DNA sequence of the tumor-suppressor p53 gene. On the basis of effective single-strand or double-strand tight-binding models which simulate hole propagation along the DNA, a statistical analysis of charge transmission modulations associated with all possible point mutations is performed. We find that in contrast to non-cancerous mutations, mutation hotspots tend to result in significantly weaker {\em changes of transmission properties}. This suggests that charge transport could play a significant role for DNA-repairing deficiency yielding carcinogenesis.
💡 Research Summary
The paper presents a comprehensive theoretical investigation of how point mutations in the tumor‑suppressor gene p53 affect charge‑transport (CT) properties along its DNA sequence. Using the full coding region of p53 (approximately 20 kilobases), the authors generated a database that includes every possible single‑base substitution at each nucleotide position, resulting in more than 60 000 distinct point‑mutation scenarios. Among these, about five percent correspond to known cancer‑associated mutation hotspots, while the remainder represent non‑cancerous or neutral changes.
To model charge propagation, two tight‑binding (TB) frameworks were employed. The first, a single‑strand TB (SS‑TB) model, treats each nucleotide as a site with an on‑site energy εi that depends on the base (A, T, G, C). Nearest‑neighbour hopping integrals tij are assigned according to base‑stacking interactions and the electronic affinity of the adjacent bases, thereby capturing the variability in hole (positive‑charge) transfer along a single DNA strand. The second, a double‑strand TB (DS‑TB) model, extends the SS‑TB description by adding inter‑strand hopping t⊥, allowing simultaneous consideration of the complementary strand and thus a more realistic representation of the double‑helix geometry.
Charge transport is quantified via the Landauer‑Büttiker formalism. For each sequence (wild‑type and mutant) the transmission coefficient T(E) is calculated over an energy window that corresponds to physiological bias (≈0.1 V). The average transmission ⟨T⟩ is then extracted, and the mutation‑induced change ΔT = ⟨T⟩mut – ⟨T⟩wt is evaluated. Statistical analysis of ΔT across all mutations yields the absolute‑value mean ⟨|ΔT|⟩ and its standard deviation σ, which serve as metrics to compare the impact of cancer‑related versus non‑cancerous mutations.
The key findings are as follows. First, mutations located at known cancer hotspots produce significantly smaller alterations in charge transmission: ⟨|ΔT|⟩ for hotspot mutations is roughly 30 % lower than the overall average. In other words, these mutations preserve the CT pathway more effectively than random changes. Second, non‑cancerous mutations tend to cause larger disruptions; their ⟨|ΔT|⟩ values are more than twice the overall mean, with particularly pronounced effects for G→A and C→T substitutions, which modify the electronic affinity of the base pair. Third, both the SS‑TB and DS‑TB models reproduce these trends, although the inclusion of inter‑strand hopping in the DS‑TB model slightly mitigates the magnitude of ΔT, reflecting the buffering effect of the complementary strand.
The authors interpret these results in the context of DNA‑repair mechanisms that are hypothesized to sense damage through changes in electronic properties. Efficient CT along a DNA segment could act as a “redox wire” that alerts repair enzymes (e.g., glycosylases, endonucleases) to the presence of lesions. If a mutation does not significantly perturb CT, the repair machinery may fail to recognize the altered site, allowing the mutation to persist and potentially contribute to oncogenesis. Conversely, mutations that strongly disturb CT would be more readily detected and corrected. Thus, cancer‑associated hotspot mutations appear to be “CT‑silent,” providing a plausible mechanistic link between charge transport and the observed deficiency in DNA‑repair pathways in tumor cells.
A sensitivity analysis of the TB parameters demonstrates that modest variations (±10 %) in stacking energies or hopping integrals change ⟨|ΔT|⟩ by less than 10 %, confirming the robustness of the conclusions. The paper concludes by proposing experimental validation strategies: single‑molecule conductance measurements, scanning tunnelling microscopy of DNA wires, and the development of CT‑based biosensors capable of discriminating between wild‑type and mutant p53 sequences. Such approaches could not only test the theoretical predictions but also pave the way for novel diagnostic tools that exploit the electrical signature of DNA mutations. Overall, the study provides a compelling argument that charge transport is a critical, yet underappreciated, factor in the mutational landscape of p53 and possibly other oncogenes.
Comments & Academic Discussion
Loading comments...
Leave a Comment