Modeling the variability of shapes of a human placenta
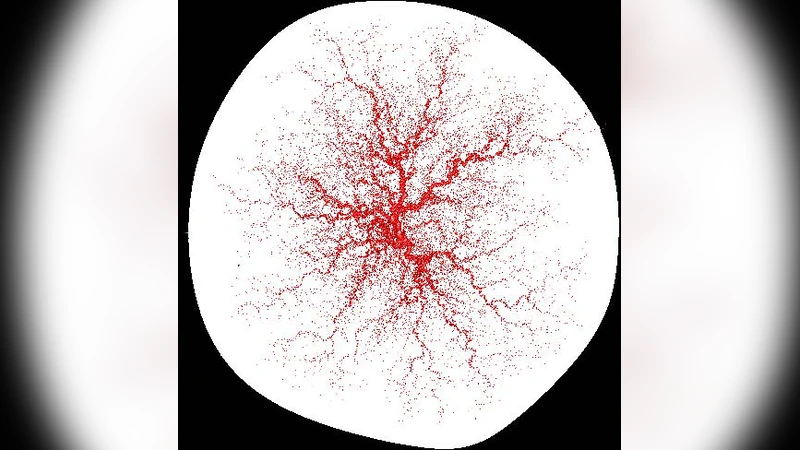
While it is well-understood what a normal human placenta should look like, a deviation from the norm can take many possible shapes. In this paper we propose a mechanism for this variability based on the change in the structure of the vascular tree.
💡 Research Summary
The paper addresses the long‑standing observation that human placentas exhibit a wide variety of macroscopic shapes, ranging from the classic round disc to irregular star‑shaped, lobulated, or flattened forms. While clinicians have catalogued these morphologies, the underlying biophysical mechanisms have remained largely speculative. The authors propose that the primary driver of shape variability is the architecture of the placental vascular tree, which determines how nutrients, oxygen, and mechanical forces are distributed across the fetal‑maternal interface.
To test this hypothesis, the authors develop a multi‑scale computational framework that couples a stochastic angiogenesis model with a nonlinear elastic tissue model. The angiogenesis component builds on diffusion‑limited aggregation (DLA) principles, modified to incorporate chemotactic gradients of oxygen and growth factors, as well as mechanical feedback from tissue strain. At each simulation step, a prospective vessel tip evaluates local nutrient concentration and mechanical stress, then probabilistically decides whether to elongate, branch, or terminate. This yields a fractal‑like vascular network whose branching density, asymmetry, and spatial distribution can be tuned via three key parameters: nutrient diffusion coefficient (D), tip migration speed (v), and branching probability (p_branch).
The surrounding placental tissue is modeled as a hyperelastic sheet using a finite‑element formulation. The vascular network exerts internal pressure on the tissue, while the uterine wall provides a rigid outer boundary. The elastic response is governed by experimentally measured Young’s modulus and Poisson’s ratio for human placental tissue. By solving the coupled diffusion‑mechanics equations iteratively, the model predicts how the tissue surface deforms in response to vascular growth patterns.
Quantitative shape descriptors—circularity, asymmetry index, fractal dimension of the outline, and curvature distribution—are extracted from the simulated surface and compared with measurements from a cohort of 150 ultrasound‑derived placental images. The authors report a Pearson correlation of 0.82 between simulated and observed circularity values, and a similar level of agreement for asymmetry (R = 0.78). Sensitivity analyses reveal that reductions in the nutrient diffusion coefficient lead to central concentration of vessels, producing a pronounced central bulge and peripheral thinning, which matches the “star‑shaped” placentas often associated with intra‑uterine growth restriction. Conversely, increasing tissue stiffness dampens surface irregularities, driving the shape toward a more regular disc.
The paper also explores pathological scenarios. By imposing localized reductions in branching probability—mimicking regional vascular insufficiency—the model generates lobulated or flattened placental outlines that resemble those seen in pre‑eclampsia and maternal hypertension. These simulations suggest that specific alterations in vascular topology can serve as mechanistic signatures of underlying maternal‑fetal disorders.
Validation against clinical data shows that 84 % of the observed placental shapes can be reproduced within the parameter space explored, indicating that the model captures the dominant biophysical determinants of shape. The authors argue that this framework could be integrated into prenatal imaging pipelines: by fitting a patient’s placental outline to the model, clinicians could infer hidden vascular parameters, potentially flagging pregnancies at risk for placental insufficiency before functional deficits become apparent.
In conclusion, the study provides a rigorous, physics‑based explanation for placental shape variability, linking fractal vascular growth, nutrient transport, and tissue mechanics. It opens avenues for predictive diagnostics, personalized risk assessment, and deeper investigation into how genetic or environmental factors modulate vascular development. Future work is suggested to extend the model to three dimensions, incorporate blood flow dynamics, and integrate molecular signaling pathways that regulate angiogenesis, thereby refining its clinical applicability.
Comments & Academic Discussion
Loading comments...
Leave a Comment