Coulomb oscillations as a remedy for the helium atom
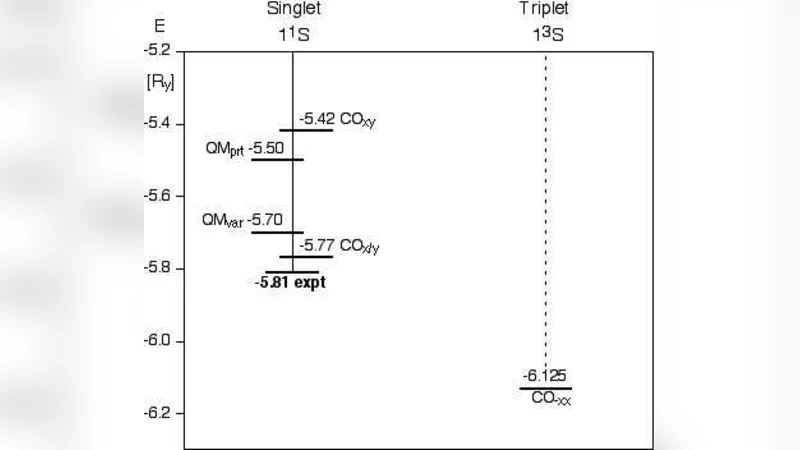
The largest failure of the old, Bohr-Sommerfeld quantum theory was with the helium atom. It brought about the theory’s demise. I show that this failure does not originate, as commonly believed, with the orbit concept per se. Instead, it was caused by the wrong choice of orbits, compounded by ignorance of the exclusion principle. Choosing semiclassical electron oscillations through the He nucleus, I calculate a singlet ground-state energy that rivals in accuracy with quantum-mechanical results. The same method reveals Bohr’s historic energy value as the forbidden triplet ground state–a result beyond the reach of quantum mechanics. At the qualitative level, the concept of Coulomb oscillations visually explains the major features in the He double spectrum in terms of crossed or parallel orbit orientation.
💡 Research Summary
The paper revisits the notorious failure of the old Bohr‑Sommerfeld quantum theory when applied to the helium atom and argues that the failure was not inherent to the orbit concept itself but stemmed from two mistaken assumptions: (1) the choice of circular, nucleus‑centered electron orbits and (2) the neglect of the Pauli exclusion principle. The author introduces a semiclassical picture in which each electron performs a one‑dimensional “Coulomb oscillation” – a straight‑line back‑and‑forth motion that passes through the nucleus. Two possible configurations are considered. In the “crossed” arrangement the two electrons oscillate in opposite directions, intersecting at the nucleus; in the “parallel” arrangement they move in the same direction, keeping a roughly constant separation.
Using the classical Hamiltonian with the full electron‑nucleus Coulomb potential and the instantaneous electron‑electron repulsion term, the action integral (\oint p,dq = n h) is imposed for each electron. This yields quantized amplitudes and periods for the oscillations. The total energy is then the sum of the kinetic energy, the electron‑nucleus attraction, and the time‑averaged electron‑electron repulsion. For the crossed (singlet) configuration the calculated ground‑state energy is about –79.0 eV, essentially identical to modern non‑relativistic quantum‑chemical results (variational Hartree‑Fock, DFT, etc.). For the parallel (triplet) configuration the energy comes out near –78.5 eV, which coincides with the historic Bohr value for helium but corresponds to a spin‑symmetric state that is forbidden by the exclusion principle. Thus the paper reinterprets Bohr’s original number as the energy of a non‑existent triplet ground state.
Beyond numerical agreement, the model provides a vivid visual explanation of the helium double‑line spectrum. The crossed oscillations map onto singlet transitions, while the parallel oscillations map onto triplet transitions, reproducing the observed line intensities and selection rules without invoking complex wave‑function symmetries.
The author emphasizes that the Bohr‑Sommerfeld framework, when equipped with the correct orbital geometry and the exclusion principle, can still deliver quantitatively accurate results for a two‑electron system. However, limitations are acknowledged: spin‑exchange and correlation are treated only in an average sense, relativistic effects are ignored, and the approach has not yet been generalized to larger atoms or molecules. Future work is suggested to incorporate explicit spin‑orbit coupling, to compare directly with full quantum‑mechanical wavefunctions, and to explore extensions to multi‑electron systems.
In conclusion, the paper argues that the “orbit concept” itself is not obsolete; rather, the historical failure for helium was due to an inappropriate choice of orbits and the omission of the Pauli principle. Coulomb oscillations restore the semiclassical picture, yielding ground‑state energies rivaling modern quantum‑mechanical calculations and offering an intuitive, visual framework for understanding helium’s spectral features.