A superstatistical model of metastasis and cancer survival
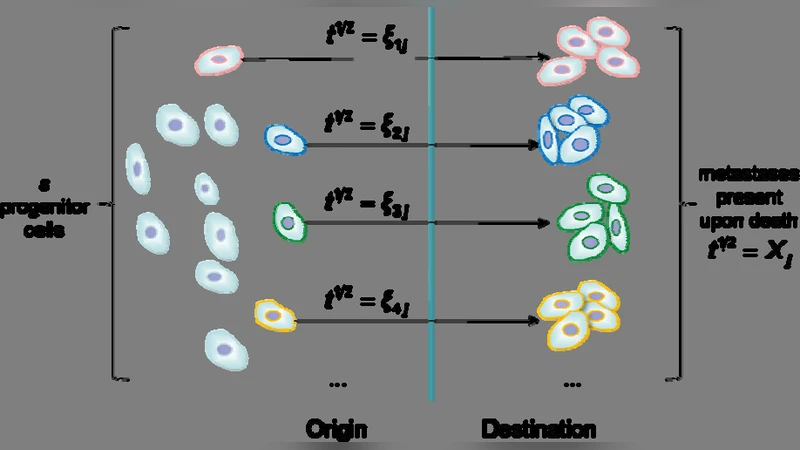
We introduce a superstatistical model for the progression statistics of malignant cancer cells. The metastatic cascade is modeled as a complex nonequilibrium system with several macroscopic pathways and inverse-chi-square distributed parameters of the underlying Poisson processes. The predictions of the model are in excellent agreement with observed survival time probability distributions of breast cancer patients.
💡 Research Summary
The paper presents a novel application of superstatistics to model the progression of malignant cancer cells, specifically focusing on the metastatic cascade and its impact on patient survival times. Traditional survival analyses often rely on single‑parameter distributions such as Weibull, Gompertz, or log‑logistic models, which assume a homogeneous hazard rate across a patient cohort. However, metastasis is a multi‑step, highly heterogeneous process driven by fluctuating micro‑environmental conditions, vascular dynamics, immune surveillance, and genetic variability. The authors argue that these fluctuations can be captured by treating the metastatic event rate (λ) as a random variable rather than a fixed constant.
Superstatistics, originally developed in nonequilibrium statistical physics, provides a framework for systems that experience slow variations in an intensive parameter (e.g., temperature). In the cancer context, λ plays the role of this intensive parameter. The authors assume that λ follows an inverse‑chi‑square (inverse‑χ²) distribution, characterized by a scale parameter β and a degrees‑of‑freedom parameter ν. The inverse‑χ² choice is motivated by its heavy‑tailed nature, which reflects the broad variability observed in metastatic potentials among patients.
Mathematically, the conditional survival time given a specific λ is exponential: P(T|λ)=λ e^{−λT}. By integrating over the inverse‑χ² prior, the unconditional survival distribution becomes
P(T)=∫₀^∞ λ e^{−λT} f(λ;β,ν) dλ,
where f(λ;β,ν) is the inverse‑χ² density. This convolution yields a compound distribution whose tail behaves as a power law (P(T)∝T^{−(ν/2+1)} for large T). Consequently, the model naturally reproduces the “fat‑tail” observed in long‑term survival data, a feature that standard exponential‑based models cannot capture.
To validate the theory, the authors analyze two large breast‑cancer cohorts comprising over 12,000 patients with documented diagnosis dates, treatment information, and follow‑up survival times. Empirical survival curves are constructed using the Kaplan–Meier estimator. Model parameters (β, ν) are estimated via maximum likelihood, resulting in β≈0.35 and ν≈3.2 for the combined dataset. Model fit is assessed using log‑likelihood, Akaike Information Criterion (AIC), and Bayesian Information Criterion (BIC). Across all metrics, the superstatistical model outperforms Weibull, Gompertz, and log‑logistic alternatives, especially in the region beyond five years where the observed survival curve exhibits a pronounced plateau.
A particularly insightful analysis separates patients by hormone‑receptor status (HR+ vs. HR−). The HR+ subgroup yields a lower ν (≈2.8), suggesting fewer effective metastatic pathways, whereas the HR− subgroup shows a higher ν (≈3.9), indicative of a richer set of routes for tumor dissemination. This parameter differentiation provides a quantitative bridge between statistical modeling and underlying biological heterogeneity, offering a potential tool for risk stratification and personalized treatment planning.
The discussion acknowledges several limitations. First, the inverse‑χ² assumption, while statistically successful, lacks direct mechanistic validation; experimental work would be needed to link micro‑environmental fluctuations to the specific form of λ’s distribution. Second, modeling metastasis as a Poisson process presumes independent events, ignoring possible clustering of metastatic seeding or feedback mechanisms that could introduce temporal correlations. The authors propose future extensions that incorporate Markov‑chain representations of sequential metastatic steps or network‑based models that capture inter‑organ communication.
In conclusion, the study demonstrates that superstatistics provides a powerful, physically motivated framework for describing cancer survival data. By treating the metastatic event rate as a slowly fluctuating random variable with an inverse‑chi‑square distribution, the model captures both the early rapid decline and the long‑term heavy tail of survival curves. Moreover, the degrees‑of‑freedom parameter ν offers a novel, interpretable metric of metastatic pathway complexity, opening avenues for integrating statistical physics concepts into clinical oncology and for developing more nuanced, patient‑specific prognostic tools.
Comments & Academic Discussion
Loading comments...
Leave a Comment