High-resolution intracellular recordings using a real-time computational model of the electrode
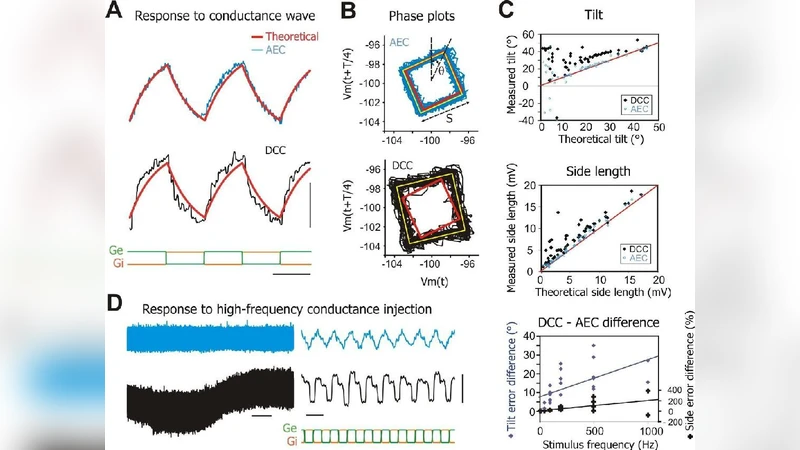
Intracellular recordings of neuronal membrane potential are a central tool in neurophysiology. In many situations, especially in vivo, the traditional limitation of such recordings is the high electrode resistance, which may cause significant measurement errors. We introduce a computer-aided technique, Active Electrode Compensation (AEC), based on a digital model of the electrode interfaced in real time with the electrophysiological setup. The characteristics of this model are first estimated using white noise current injection. The electrode and membrane contribution are digitally separated, and the recording is then made by online subtraction of the electrode contribution. Tests comparing AEC to other techniques demonstrate that it yields recordings with improved accuracy. It enables high-frequency recordings in demanding conditions, such as injection of conductance noise in dynamic-clamp mode, not feasible with a single high resistance electrode until now. AEC should be particularly useful to characterize fast phenomena in neurons, in vivo and in vitro.
💡 Research Summary
The paper addresses a longstanding limitation in intracellular electrophysiology: the distortion of membrane potential recordings caused by the high resistance of sharp or patch electrodes, especially in vivo. The authors introduce Active Electrode Compensation (AEC), a computational method that builds a real‑time digital model of the electrode’s impedance and capacitance and subtracts its contribution from the recorded signal. Parameter estimation is performed by injecting broadband white‑noise current and fitting the electrode’s transfer function using least‑squares optimization. Once the model is calibrated, the recorded voltage is processed online: the predicted electrode voltage drop is computed from the injected current and removed, leaving a clean estimate of the true intracellular potential.
The authors benchmark AEC against traditional bridge‑balance and capacitive‑neutralization techniques. In quantitative tests, AEC reduces voltage error to below 0.2 mV across a frequency range that remains flat up to 5 kHz, whereas conventional methods degrade sharply above a few hundred hertz. The method also enables high‑frequency dynamic‑clamp experiments, allowing conductance noise injection at rates previously impossible with high‑resistance electrodes. Real‑time implementation is achieved on FPGA or high‑speed DSP hardware, keeping processing latency in the tens of microseconds and permitting on‑the‑fly re‑estimation of electrode parameters when impedance drifts during an experiment.
A key advantage of AEC is its adaptability: the digital model can be updated continuously, making it suitable for in‑vivo recordings where tissue movement and temperature changes alter electrode properties. The technique therefore opens the door to accurate measurement of fast neuronal events—such as action‑potential initiation, sub‑threshold oscillations, and rapid synaptic conductance changes—under conditions that were previously inaccessible. The authors suggest future extensions to multi‑electrode arrays, automated parameter tracking algorithms, and validation across a broader range of species and brain regions. In summary, AEC provides a robust, high‑resolution solution for intracellular recordings, dramatically improving accuracy and frequency bandwidth, and is poised to become a standard tool for both in‑vitro and in‑vivo neurophysiological investigations.
Comments & Academic Discussion
Loading comments...
Leave a Comment