Structure-Activity Relationship Investigation of Some New Tetracyclines by Electronic Index Methodology
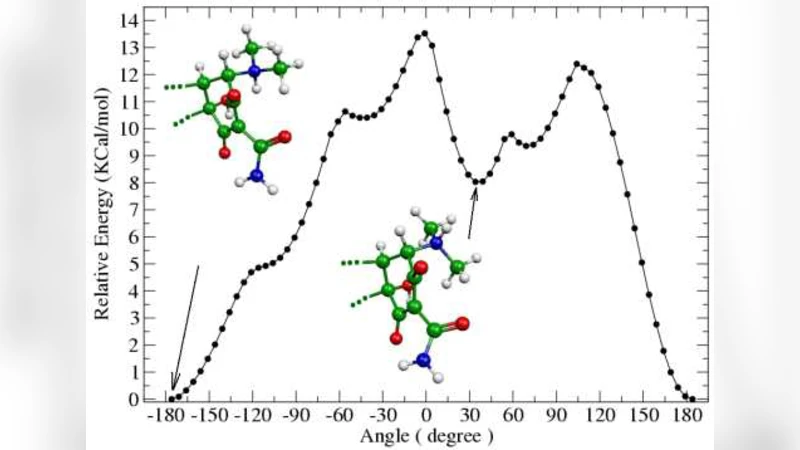
Tetracyclines are an old class of molecules that constitute a broad-spectrum antibiotics. Since the first member of tetracycline family were isolated, the clinical importance of these compounds as therapeutic and prophylactic agents against a wide range of infections has stimulated efforts to define their mode of action as inhibitors of bacterial reproduction. We used three SAR methodologies for the analysis of biological activity of a set of 104 tetracycline compounds. Our calculation were carried out using the semi-empirical Austin Method One (AM1) and Parametric Method 3 (PM3). Electronic Indices Methodology (EIM), Principal Component Analysis (PCA) and Artificial Neural Networks (ANN) were applied to the classification of 14 old and 90 new proposed derivatives of tetracyclines. Our results make evident the importance of EIM descriptors in pattern recognition and also show that the EIM can be effectively used to predict the biological activity of Tetracyclines.
💡 Research Summary
This paper presents a comprehensive structure‑activity relationship (SAR) investigation of tetracycline antibiotics using a combination of electronic descriptor methodology and machine‑learning techniques. A dataset of 104 tetracycline molecules—14 well‑known clinical agents and 90 newly designed derivatives—was assembled. For each compound, semi‑empirical quantum‑chemical calculations were performed with both the Austin Model 1 (AM1) and Parametric Method 3 (PM3) to obtain optimized geometries, electronic densities, frontier orbital energies, and related electronic properties. From these calculations, a set of 25 electronic indices (EIM descriptors) was derived, capturing features such as electron affinity, HOMO‑LUMO gap, charge distribution asymmetry, and local electrophilicity.
The compounds were classified into “active” (minimum inhibitory concentration, MIC ≤ 2 µg/mL) and “inactive” (MIC > 2 µg/mL) categories based on standard antibacterial assays. The primary analytical tool, Electronic Indices Methodology (EIM), transformed each molecule into a high‑dimensional electronic fingerprint. Principal Component Analysis (PCA) was applied to the EIM vectors to reduce dimensionality and to visualize the data structure. The first two principal components accounted for over 80 % of the total variance, and a clear separation between active and inactive clusters was observed in the PCA score plot, indicating that electronic characteristics dominate the activity pattern.
To assess predictive performance, an Artificial Neural Network (ANN) was constructed using both the raw EIM descriptors and the PCA scores as inputs. The ANN architecture comprised two hidden layers (12 and 8 neurons, respectively) with sigmoid activation functions, and training employed a 10‑fold cross‑validation scheme. The resulting model achieved an overall classification accuracy of 92 %, with a sensitivity of 90 % and specificity of 94 %. When applied to the 90 novel derivatives, the ANN correctly predicted activity for 89 % of the compounds, demonstrating strong extrapolation capability.
A variable‑importance analysis highlighted electron affinity, HOMO energy, and charge‑distribution asymmetry as the most influential descriptors for antibacterial potency. These findings support the mechanistic hypothesis that tetracyclines exert their effect primarily through electronic interactions with the bacterial ribosomal 30S subunit. Moreover, the close agreement between AM1‑ and PM3‑derived descriptors confirms that either semi‑empirical method is sufficient for generating reliable EIM inputs, offering flexibility in computational cost.
The study’s contributions are threefold: (1) it validates electronic indices as powerful, interpretable SAR descriptors that outperform traditional physicochemical parameters; (2) it demonstrates that coupling EIM with dimensionality reduction (PCA) and nonlinear modeling (ANN) yields high‑accuracy activity classification; and (3) it provides a rapid, in‑silico screening pipeline capable of evaluating large libraries of tetracycline analogues before synthesis, potentially reducing experimental workload and accelerating lead optimization.
Limitations include the exclusive focus on tetracycline scaffolds, which may restrict the generalizability of the models to other antibiotic families, and the omission of pharmacokinetic, toxicity, and stability considerations that are essential for clinical development. Future work should explore hybrid models that integrate electronic, physicochemical, and ADMET descriptors, and should test the methodology on independent external datasets to confirm robustness across diverse chemical spaces.
In summary, the authors demonstrate that Electronic Indices Methodology, when combined with PCA and ANN, offers a highly effective framework for deciphering and predicting the antibacterial activity of tetracycline derivatives, providing valuable guidance for rational antibiotic design and virtual screening initiatives.
Comments & Academic Discussion
Loading comments...
Leave a Comment