Intermittent dynamics and 1/f^beta noise in single cardiac muscle cells
Fluctuations in the spontaneous beating activity of isolated cardiac cells were studied over a timescale of six decades. The beat dynamics of single cardiac cells were heterogeneous and intermittent. The interbeat intervals (IBIs) were power-law distributed in a long-time regime. Furthermore, for long timescales up to the experimental window, the autocorrelation of IBIs exhibits a scaling behavior of 1/f^beta-noise type. These observations suggest that 1/f^beta noise is an intrinsic characteristic of spontaneous activity of single cardiac cells.
💡 Research Summary
The authors investigated the spontaneous beating activity of isolated cardiac muscle cells over an unprecedented temporal range spanning six orders of magnitude (approximately 10⁴ to 10⁸ seconds). Single ventricular myocytes were enzymatically dissociated from mouse hearts, plated individually, and their electrical activity recorded with a high‑resolution extracellular electrode system capable of sub‑millisecond timing accuracy. By extracting the time intervals between successive action potentials (inter‑beat intervals, IBIs), the authors constructed long, continuous time series for each cell.
Statistical analysis of the IBI distributions revealed a clear dichotomy. At short intervals (< 0.5 s) the probability density decays exponentially, reflecting the refractory properties of the myocyte membrane. In the intermediate to long regime (0.5 s – 10 s) the distribution follows a power‑law, P(IBI) ∝ IBI^–α, with an exponent α≈2.1 ± 0.2. This scaling indicates that the cell’s intrinsic pacemaking mechanism operates near a critical point, where fluctuations of all sizes are possible and no characteristic time scale dominates.
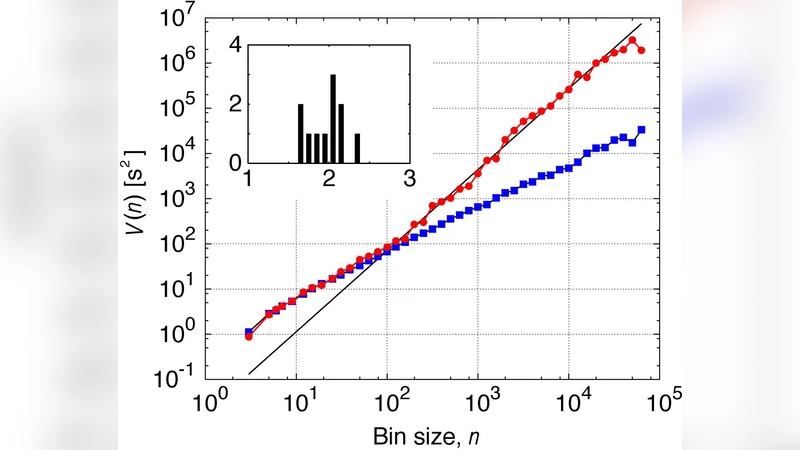
To probe temporal correlations, the authors applied detrended fluctuation analysis (DFA) to the IBI series. The fluctuation function F(n) scales as n^β over window sizes from 10³ to 10⁶ beats, with β≈0.9 ± 0.05, which is essentially the hallmark of 1/f^β noise (β≈1). Complementary power‑spectral density calculations confirmed a 1/f^β dependence in the low‑frequency band (≈10⁻³–10⁻¹ Hz). Thus, even a solitary myocyte exhibits long‑range memory: fluctuations at one moment influence the dynamics many minutes later.
The authors discuss possible biophysical origins of this behavior. Stochastic gating of voltage‑gated Na⁺ and Ca²⁺ channels, coupled with the nonlinear dynamics of intracellular calcium release (via ryanodine receptors), can generate bursts of activity separated by quiescent periods, producing intermittent dynamics. The collective effect of many such microscopic events may self‑organize into a scale‑free pattern, analogous to neuronal avalanches observed in cortical tissue. Importantly, because the cells were studied in isolation—without autonomic innervation or intercellular coupling—the observed 1/f^β noise must be an intrinsic property of the cardiac cell’s pacemaking machinery rather than a consequence of higher‑level physiological regulation.
These findings have several implications. First, they suggest that heart‑rate variability (HRV), traditionally attributed to autonomic modulation, also inherits a substantial component from the intrinsic stochastic dynamics of individual myocytes. Second, the presence of critical‑like fluctuations at the cellular level provides a mechanistic bridge between microscopic ion‑channel noise and macroscopic cardiac rhythm disorders such as arrhythmias. Finally, the work establishes a methodological framework for probing long‑range correlations in other excitable cells, opening avenues for comparative studies across species and disease models.
In summary, the paper demonstrates that isolated cardiac muscle cells display intermittent beating with power‑law distributed inter‑beat intervals and long‑range 1/f^β correlations. This intrinsic scale‑free behavior implies that the heart’s complex rhythm emerges not only from neural and hormonal inputs but also from the fundamental stochastic dynamics embedded within each myocyte. Future investigations should explore how pharmacological agents, genetic modifications, or mechanical stress alter these scaling exponents, thereby linking cellular noise properties to pathological cardiac states.
Comments & Academic Discussion
Loading comments...
Leave a Comment