Shape instabilities in vesicles: a phase-field model
A phase field model for dealing with shape instabilities in fluid membrane vesicles is presented. This model takes into account the Canham-Helfrich bending energy with spontaneous curvature. A dynamic equation for the phase-field is also derived. With this model it is possible to see the vesicle shape deformation dynamically, when some external agent instabilizes the membrane, for instance, inducing an inhomogeneous spontaneous curvature. The numerical scheme used is detailed and some stationary shapes are shown together with a shape diagram for vesicles of spherical topology and no spontaneous curvature, in agreement with known results.
💡 Research Summary
The paper introduces a phase‑field framework to study shape instabilities of fluid‑membrane vesicles, incorporating the classic Canham‑Helfrich bending energy together with a spatially varying spontaneous curvature. Traditional approaches treat the vesicle surface as a sharp interface and require explicit curvature calculations, which become cumbersome when the membrane is driven out of equilibrium by external agents. By defining a scalar order parameter φ(x,t) that distinguishes interior (φ≈+1) from exterior (φ≈−1) regions, the authors embed the vesicle in a three‑dimensional diffuse interface. The free‑energy functional consists of two parts: (i) a double‑well potential W(φ)=¼(φ²−1)² and a gradient term (ε²/2)|∇φ|² that enforce a thin interfacial layer of thickness ε, and (ii) a curvature contribution that reproduces the Helfrich term (κ/2)(2H−C₀)² through combinations of ∇φ and ∇²φ. The spontaneous curvature C₀(x) can be prescribed arbitrarily, allowing the model to mimic heterogeneous protein adsorption, lipid composition gradients, or external fields.
The dynamics follow a conserved Cahn‑Hilliard equation: ∂ₜφ = M∇²μ, where μ = δF/δφ is the chemical potential and M is a mobility coefficient. This formulation guarantees volume conservation and monotonic decrease of the total free energy. For numerical implementation the authors employ a regular cubic lattice, central finite differences for spatial derivatives, and a semi‑implicit Crank‑Nicolson time stepping scheme. The interface thickness ε is chosen on the order of two lattice spacings to balance accuracy and computational cost, while the mobility M is tuned to achieve reasonable physical time scales. A Lagrange multiplier enforces the global volume constraint, and the initial condition is a spherical vesicle with φ=+1 inside.
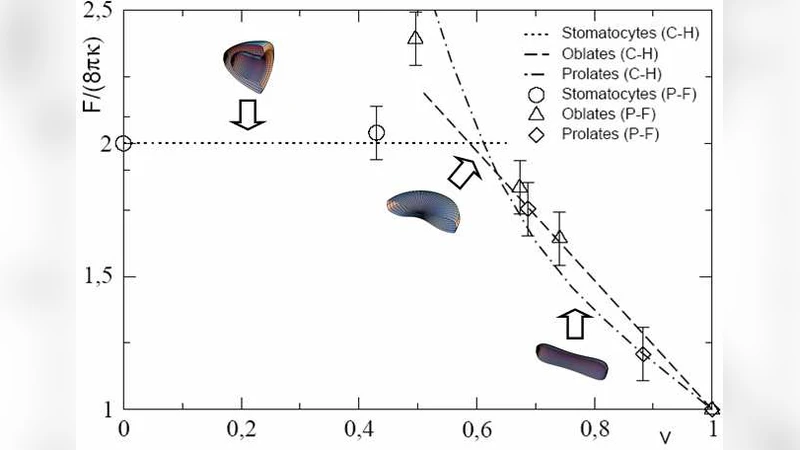
Two families of simulations are presented. In the first set, C₀ is set to zero everywhere, and the reduced volume v (the ratio of the vesicle volume to that of a sphere with the same surface area) is varied. The model reproduces the well‑known sequence of equilibrium shapes: spheres at v≈1, prolate ellipsoids for intermediate v, and stomatocyte‑like or “spindle” shapes at lower v. The computed shape diagram matches analytical predictions and previous surface‑evolver studies, confirming that the phase‑field approach captures the correct energetics of curvature‑driven shape transitions.
In the second set, the authors impose a non‑uniform spontaneous curvature, for example a positive C₀ on one hemisphere and a negative C₀ on the opposite side. The vesicle then undergoes a time‑dependent deformation toward asymmetric configurations such as pear‑shaped or budded forms. The evolution of φ’s isosurfaces and the associated curvature energy density are visualized, providing insight into how localized curvature cues propagate and reshape the membrane. These results demonstrate that the model can dynamically follow shape changes triggered by external agents, something that static energy minimization methods cannot achieve.
The discussion highlights several advantages of the phase‑field formulation: (1) external fields or heterogeneous material properties can be incorporated simply by modifying C₀(x) or adding additional free‑energy terms; (2) topological changes (e.g., vesicle fission or fusion) are naturally handled because the interface is diffuse; (3) the method is amenable to parallel computation and can be extended to three‑dimensional simulations with modest computational resources. Limitations are also acknowledged. The diffuse interface thickness ε is larger than the actual bilayer thickness, which may smear fine‑scale features; the results can be sensitive to the choice of mobility M and the discretization parameters; and quantitative comparison with experiments requires careful calibration of κ, C₀, and the mapping between simulation time and physical time.
Future work suggested includes coupling the phase‑field vesicle model to hydrodynamic flow (e.g., using a Navier‑Stokes solver), incorporating explicit protein–membrane interactions, and performing multiscale simulations where ε is reduced locally near regions of high curvature. Overall, the paper provides a robust, versatile computational tool for exploring the rich landscape of vesicle shape instabilities, bridging the gap between static curvature theory and fully dynamic, heterogeneous membrane phenomena.
Comments & Academic Discussion
Loading comments...
Leave a Comment