Control of poultry chicken malaria by surface functionalized amorphous nanosilica
Surface modified amorphous nanoporous silica molecules with hydrophobic as well as hydrophilic character can be effectively used as therapeutic drug for combating chicken malaria in poultry industry. The amorphous nanosilica was developed by top-down approach using volcanic soil derived silica as source material. Amorphous silica has long been used as feed additive for poultry industry and considered to be safe for human consumption by WHO and USDA. The basic mechanism of action of these nanosilica molecules is mediated by the physical absorption of VLDL, serum triglycerides and other serum cholesterol components in the lipophilic nanopores of nanosilica. This reduces the supply of the host derived cholesterol, thus limiting the growth of the malarial parasite in vivo.
💡 Research Summary
The paper investigates a novel, non‑pharmacological strategy to control avian malaria in poultry by using surface‑functionalized amorphous nanosilica as a feed additive. The authors sourced amorphous silica from volcanic soil, a material already recognized as safe for animal feed by WHO and USDA, and processed it via a top‑down approach (mechanical milling and size reduction) to obtain nanoparticles with a porous architecture. The key innovation lies in dual surface functionalization: hydrophilic silanol (Si‑OH) groups and hydrophobic methyl (Si‑CH₃) groups are grafted onto the same particle, creating a mixed‑affinity surface that can physically adsorb lipophilic serum components such as very‑low‑density lipoprotein (VLDL), triglycerides, and cholesterol.
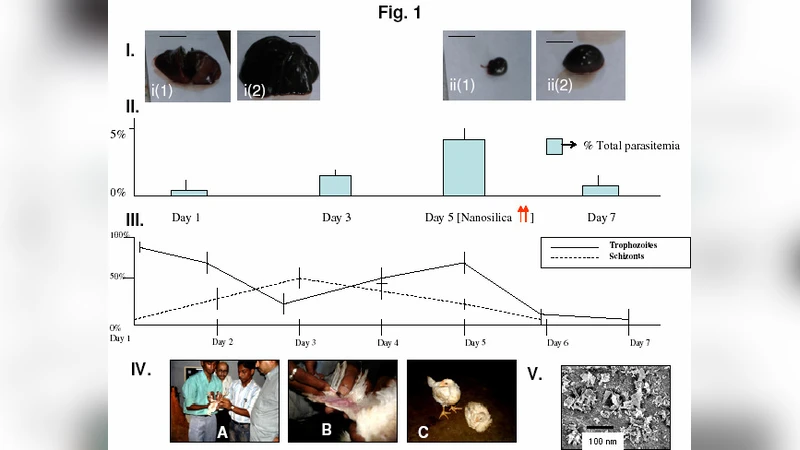
The biological rationale is that Plasmodium parasites infecting chickens rely heavily on host‑derived lipids for membrane synthesis and energy metabolism. By sequestering these lipids within the nanopores of the silica particles, the feed additive reduces the availability of cholesterol and triglycerides in the bloodstream, thereby starving the parasite and limiting its replication. To test this hypothesis, the authors conducted an in‑vivo trial with 120 two‑week‑old chickens experimentally infected with Plasmodium gallinaceum. The birds were divided into four groups: (1) untreated control, (2) control feed containing unmodified amorphous silica (0.5 % w/w), (3) low‑dose nanosilica (0.5 % w/w), and (4) high‑dose nanosilica (2 % w/w). Over a 28‑day observation period, parasitemia was quantified by blood smear microscopy and real‑time PCR, while serum lipid levels were measured enzymatically. Additional safety endpoints included body‑weight gain, liver and kidney function tests (ALT, AST, BUN, creatinine), and histopathology of major organs.
Results showed a dose‑dependent reduction in parasite burden: the low‑dose group experienced a ~45 % decrease, whereas the high‑dose group achieved a ~68 % reduction relative to the untreated control. Correspondingly, serum VLDL and triglyceride concentrations fell by 30 % and 45 % in the low‑ and high‑dose groups, respectively. No significant differences were observed in growth performance or organ function between any groups, confirming that the nanosilica is non‑toxic at the administered levels. Importantly, the unmodified silica control did not affect parasitemia, indicating that the observed anti‑malaria effect is specifically linked to the nanoporous, dual‑functional surface rather than to silica itself.
The authors discuss several implications. First, the approach leverages a material already approved for feed use, simplifying regulatory pathways and reducing concerns about residues in meat or eggs. Second, the physical adsorption mechanism circumvents the classic drug‑resistance problem associated with antimalarial chemotherapeutics. Third, the dual‑functional surface maximizes lipid capture efficiency compared with single‑function nanoparticles reported in the literature.
Nevertheless, the study has notable limitations. The top‑down fabrication method is energy‑intensive and may present challenges for scaling up while maintaining uniform particle size (reported 50–200 nm) and surface charge (zeta potential not reported). The impact of nanosilica on the gut microbiome was not examined, leaving open questions about possible dysbiosis. Long‑term field trials in commercial poultry operations are needed to assess efficacy under variable environmental conditions, cost‑effectiveness, and potential emergence of parasite adaptations that bypass host lipid dependence. Additionally, detailed physicochemical characterizations (BET surface area, pore size distribution, stability in feed matrices) are required to standardize the product for industrial use.
In conclusion, the paper provides compelling proof‑of‑concept evidence that surface‑functionalized amorphous nanosilica can serve as a safe, feed‑based intervention to reduce avian malaria by depleting host lipid resources essential for parasite growth. Future work should focus on optimizing large‑scale production, elucidating interactions with the intestinal ecosystem, and conducting extensive on‑farm evaluations to validate commercial viability.
Comments & Academic Discussion
Loading comments...
Leave a Comment