Computational investigations into the orgins of short term biochemical memory in T cell activation
Recent studies have reported that T cells can integrate signals between interrupted encounters with Antigen Presenting Cells (APCs) in such a way that the process of signal integration exhibits a form of memory. Here, we carry out a computational study using a simple mathematical model of T cell activation to investigate the ramifications of interrupted T cell-APC contacts on signal integration. We consider several mechanisms of how signal integration at these time scales may be achieved and conclude that feedback control of immediate early gene products (IEGs) appears to be a highly plausible mechanism that allows for effective signal integration and cytokine production from multiple exposures to APCs. Analysis of these computer simulations provides an experimental roadmap involving several testable predictions.
💡 Research Summary
The paper addresses a recently observed phenomenon in immunology: T cells can retain a short‑term biochemical “memory” that allows them to integrate signals from multiple, interrupted encounters with antigen‑presenting cells (APCs). To explore how such memory might arise on the time scale of minutes to tens of minutes, the authors construct a minimal deterministic model of T‑cell activation. The model captures four core processes: (1) T‑cell receptor (TCR) engagement leading to rapid calcium influx and MAPK/ERK activation; (2) induction of immediate‑early genes (IEGs) such as c‑Fos and c‑Jun; (3) a positive feedback loop in which IEG protein products enhance their own transcription and prime downstream transcription factors; and (4) production of the effector cytokine IL‑2.
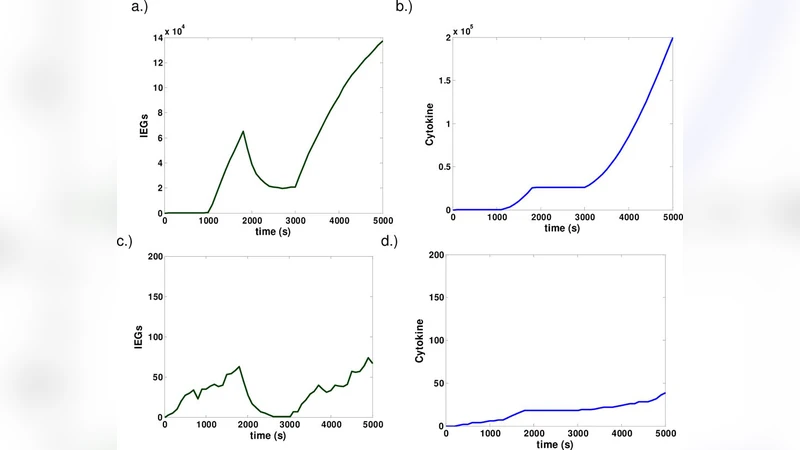
Using ordinary differential equations, the authors simulate two discrete APC contacts separated by a variable interval Δt (0–60 min). They test four candidate mechanisms for signal persistence: simple exponential decay of the TCR signal, sustained activity of kinases/phosphatases, calcium store refilling, and IEG‑mediated feedback. Parameter values are drawn from published kinetic data, and sensitivity analyses are performed to identify the most influential parameters.
Results show that simple decay and kinase persistence fail to preserve information beyond ~10 min; calcium refilling provides only a modest memory that disappears once calcium equilibrates. In contrast, the IEG feedback architecture maintains a robust memory across a broad Δt window (5–30 min). When the second APC encounter occurs within this window, IL‑2 transcription is amplified 2.5‑ to 3‑fold relative to a single encounter, matching experimental observations of “integrated” cytokine output. The model identifies three key determinants of memory strength: the transcription rate of IEGs (k_trans), the protein half‑life of IEG products (τ_IEG), and the feedback gain (α_feedback). Reducing k_trans or shortening τ_IEG abolishes memory, while excessive α_feedback leads to over‑amplification and potential cytokine storm‑like behavior.
Based on these computational insights, the authors propose a concrete experimental roadmap. First, they suggest live‑cell imaging of fluorescent IEG reporters to quantify nuclear accumulation as a function of Δt. Second, they recommend siRNA‑mediated knockdown or CRISPR activation of IEGs to modulate feedback strength and assess consequent IL‑2 output. Third, pharmacological inhibition of calcium or MAPK pathways would serve as controls to demonstrate the specificity of the IEG mechanism. Finally, microfluidic single‑cell platforms could precisely control T‑cell–APC contact timing, allowing direct validation of the predicted memory window.
In summary, the study argues that a transcription‑level feedback loop involving immediate‑early gene products provides a biologically plausible and experimentally testable mechanism for short‑term biochemical memory in T‑cell activation. This mechanism reconciles the need for rapid response (through fast signaling cascades) with the ability to integrate temporally fragmented antigenic cues. The work not only advances our theoretical understanding of T‑cell signaling dynamics but also opens new avenues for therapeutic manipulation of immune memory, such as designing vaccines or immunotherapies that exploit or modulate IEG‑mediated feedback.
Comments & Academic Discussion
Loading comments...
Leave a Comment