Two computational regimes of a single-compartment neuron separated by a planar boundary in conductance space
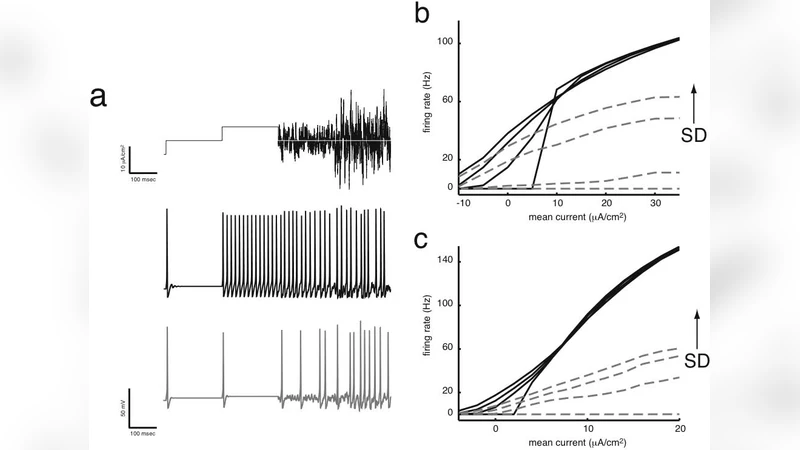
Recent in vitro data show that neurons respond to input variance with varying sensitivities. Here, we demonstrate that Hodgkin-Huxley (HH) neurons can operate in two computational regimes, one that is more sensitive to input variance (differentiating) and one that is less sensitive (integrating). A boundary plane in the 3D conductance space separates these two regimes. For a reduced HH model, this plane can be derived analytically from the V nullcline, thus suggesting a means of relating biophysical parameters to neural computation by analyzing the neuron’s dynamical system.
💡 Research Summary
In this paper the authors investigate how a single‑compartment Hodgkin‑Huxley (HH) neuron can operate in two distinct computational regimes that differ in their sensitivity to input variance. Using stochastic current injection modeled as Gaussian white noise with controllable mean (μ) and standard deviation (σ), they quantify the neuron’s output firing rate r(μ,σ) and compute its partial derivatives ∂r/∂μ (mean sensitivity) and ∂r/∂σ (variance sensitivity). When ∂r/∂σ is small relative to ∂r/∂μ the neuron behaves as an “integrator”, essentially averaging its input; when ∂r/∂σ dominates, the neuron acts as a “differentiator”, responding preferentially to rapid fluctuations.
The key discovery is that the transition between these regimes is governed by a planar boundary in the three‑dimensional conductance space defined by the maximal sodium conductance (g_Na), maximal potassium conductance (g_K), and leak conductance (g_L). By systematically varying these three parameters in extensive numerical simulations, the authors observe a sharp switch in variance sensitivity that aligns with a flat surface separating an “integrator region” from a “differentiator region”.
To understand why this surface is planar, the authors construct a reduced HH model in which the fast sodium activation and delayed potassium activation are collapsed into a single effective voltage‑dependent excitatory current and a single inhibitory current, respectively, while the leak remains linear. In this reduced system the voltage nullcline (the curve where dV/dt = 0) can be expressed analytically. The authors show that the critical condition for the neuron to change its computational mode corresponds to the nullcline becoming tangent to the voltage axis. This tangency condition yields a linear relationship among the conductances: \