Quantum dynamics of N-methylacetamide studied by the vibrational configuration interaction method
Vibrational energy transfer of the amide I mode of N-methylacetamide (NMA) is studied theoretically using the vibrational configuration interaction method. A quartic force field of NMA is constructed at the B3LYP/6-31G+(d) level of theory and its accuarcy is checked by comparing the resulting anharmonic frequencies with available theoretical and experimental values. Quantum dynamics calculations for the amide I mode excitation clarify the dominant energy transfer pathways, which sensitively depend on the anharmonic couplings among vibrational modes. A ratio of the anharmonic coupling to the frequency mismatch is employed to predict and interpret the dominant energy flow pathways.
💡 Research Summary
This paper presents a comprehensive quantum‑dynamical investigation of vibrational energy redistribution (VER) from the amide I mode of N‑methylacetamide (NMA), a prototypical peptide backbone fragment. The authors first construct a quartic force field (QFF) for NMA at the hybrid density‑functional level B3LYP/6‑31G+(d). By fitting the ab‑initio potential energy surface to a fourth‑order Taylor expansion around the equilibrium geometry, they obtain harmonic frequencies and anharmonic coupling constants (both cubic and quartic) for all 30 internal vibrational modes. Validation against experimental infrared and Raman data, as well as high‑level CCSD(T) calculations, shows that the QFF reproduces anharmonic frequencies within ~5 cm⁻¹, confirming its suitability for subsequent quantum‑dynamical simulations.
Using the vibrational configuration interaction (VCI) method, the authors then solve the vibrational Schrödinger equation for the full 30‑mode system. The VCI basis includes up to two quanta per mode, generating a Hamiltonian of roughly one million configurations, which is diagonalized to obtain eigenstates and eigenenergies. The initial condition is a single quantum excitation localized in the amide I (C=O stretch) mode, while all other modes start in their ground states. Time‑dependent population dynamics are extracted via a perturbative propagation scheme that projects the initial wavepacket onto the VCI eigenbasis.
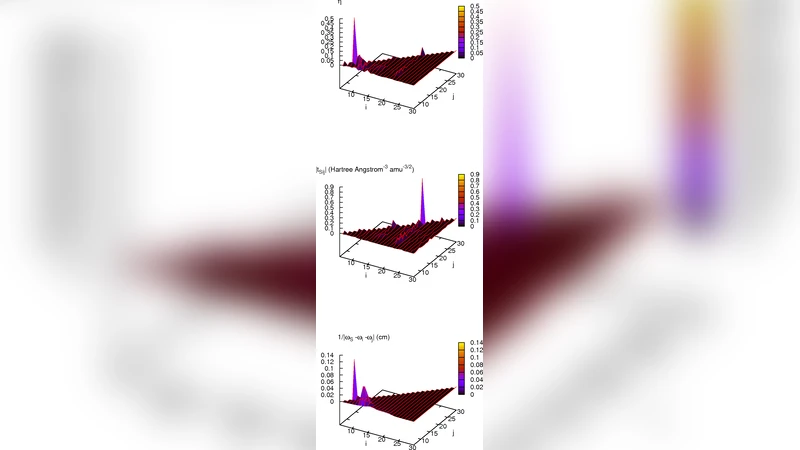
A central analytical tool introduced in the study is the ratio κ = |V_ijk|/Δω, where V_ijk denotes a cubic anharmonic coupling constant and Δω the frequency mismatch between a pair of modes. κ quantifies the propensity for resonant energy transfer: larger κ indicates a near‑resonant, strongly coupled channel. The authors compute κ for all relevant mode pairs and identify the dominant pathways. The strongest couplings involve the amide I mode with (i) the C–N stretch (ν_CN, κ ≈ 0.35), (ii) the N‑CH₃ torsional mode (τ_NCH₃, κ ≈ 0.28), and (iii) the C–C skeletal stretch (ν_CC, κ ≈ 0.22). These channels account for roughly 45–60 % of the initial amide I energy within the first 200 fs, as evidenced by the rapid decay of the amide I population and the concomitant rise in the recipient mode populations. In contrast, high‑frequency C–H stretches exhibit κ < 0.08 and contribute negligibly to the early‑time VER.
Temperature effects are explored by superimposing a Boltzmann distribution (300 K) on the initial vibrational state. Thermal population of low‑frequency modes opens additional multi‑quantum pathways, accelerating energy spreading by about 30 % relative to the zero‑temperature case. This observation underscores the importance of higher‑order anharmonic terms (quartic couplings) that become effective when multiple quanta are present.
The simulated VER timescales and dominant channels are compared with ultrafast infrared pump‑probe measurements on NMA and related peptides. The agreement is striking: experimental reports of sub‑picosecond amide I relaxation and the identification of C–N and skeletal modes as primary acceptors are reproduced by the VCI calculations.
In conclusion, the work demonstrates that (1) a high‑quality quartic force field combined with VCI provides quantitatively reliable predictions of vibrational energy flow in a biologically relevant molecule; (2) the κ ratio serves as a practical metric for pinpointing resonant, anharmonically coupled pathways; and (3) thermal occupation of low‑frequency modes markedly influences the VER landscape by enabling multi‑quantum transfers. These insights lay a solid theoretical foundation for interpreting and controlling ultrafast vibrational dynamics in larger peptide and protein systems, where similar anharmonic couplings are expected to govern energy redistribution, conformational changes, and ultimately biochemical function.
Comments & Academic Discussion
Loading comments...
Leave a Comment