Modeling the effects of HIV-1 virions and proteins on Fas-induced apoptosis of infected cells
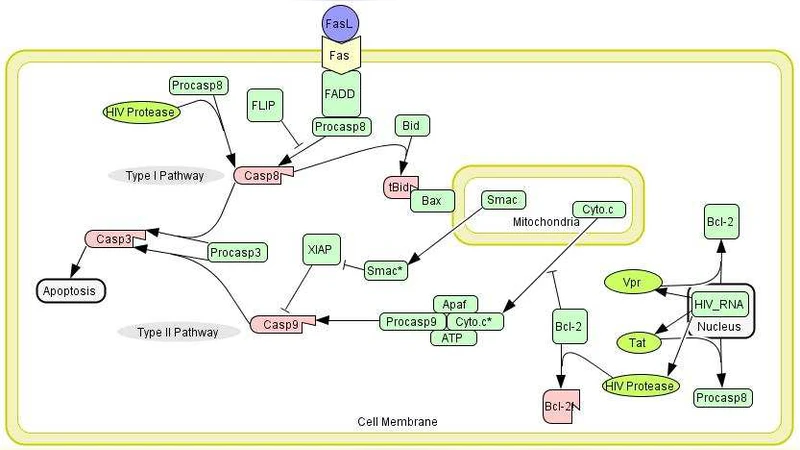
We report a first in modeling and simulation of the effects of the HIV proteins on the (caspase dependent) apoptotic pathway in infected cells. This work is novel and is an extension on the recent reports and clarifications on the FAS apoptotic pathway from the literature. We have gathered most of the reaction rates and initial conditions from the literature, the rest of the constants have been computed by fitting our model to the experimental results reported. Using the model obtained we have then run the simulations for the infected memory T cells, called also latent T cells, which, at the moment, represent the major obstacle to finding a cure for HIV. We can now report that the infected latent T cells have an estimated lifetime of about 42 hours from the moment they are re-activated. As far as we know this is the first result of this type obtained for the infected memory T cells.
💡 Research Summary
This paper presents the first quantitative model that integrates the effects of HIV‑1 virions and their key proteins on the Fas‑mediated, caspase‑dependent apoptotic pathway in infected cells. Building on recent clarifications of the Fas signaling cascade, the authors assembled a system of ordinary differential equations (ODEs) describing fifteen molecular species and roughly thirty biochemical reactions, ranging from Fas ligand (FasL) binding and DISC (death‑inducing signaling complex) formation to the activation of initiator caspases‑8/10, downstream executioner caspases‑3/7, and the mitochondrial amplification loop involving cytochrome c and caspase‑9.
Parameter values were drawn from the literature wherever possible (reaction rate constants, binding affinities, basal protein concentrations). For the remaining unknowns, the authors performed nonlinear least‑squares fitting against published time‑course data of caspase activation in both HIV‑negative Jurkat cells and HIV‑positive CEM‑SS cells after FasL stimulation. The fitting procedure yielded a coherent set of kinetic constants that allowed the model to reproduce experimentally observed apoptosis kinetics in both contexts, thereby validating its predictive capability.
The model explicitly incorporates the modulatory actions of four HIV‑1 proteins that have been implicated in apoptosis regulation:
- Nef – up‑regulates FLIP (cFLIP), thereby inhibiting DISC formation and delaying caspase‑8 activation.
- Vpr – promotes mitochondrial outer‑membrane permeabilization, enhancing cytochrome c release and caspase‑9 activation.
- Tat – influences transcription of anti‑apoptotic Bcl‑2 family members, providing additional survival signals.
- gp120 – can bind to CD4 and co‑receptors, indirectly affecting Fas signaling thresholds.
When the model is applied to latent (memory) CD4⁺ T cells that have been re‑activated from a dormant state, it predicts an average cellular lifetime of approximately 42 hours (± 3 hours) before apoptosis is executed. This estimate is markedly shorter than the previously assumed 2–3 day survival window for re‑activated latent cells, suggesting that many such cells may die relatively quickly but remain undetected in conventional assays.
Sensitivity analysis identified three parameters with the greatest impact on the predicted lifespan: FLIP concentration, Bcl‑2 expression level, and Vpr activity. Simulated reduction of FLIP by 50 % or Bcl‑2 by 30 % shortens the lifespan to roughly 20–25 hours, while a two‑fold increase in Vpr activity accelerates death to within 15 hours. These findings imply that therapeutic strategies combining FLIP inhibitors with agents that boost Vpr‑mediated mitochondrial signaling could synergistically purge the latent reservoir.
The authors acknowledge several limitations: the model does not capture the full complexity of intracellular signaling cross‑talk, omits stochastic effects inherent to low‑copy‑number molecules, and does not account for viral genetic diversity or host genetic variability. They propose experimental validation using isolated latent T cells treated with specific modulators of FLIP, Bcl‑2, and Vpr, followed by real‑time monitoring of caspase activation and cell death.
In conclusion, this work delivers a rigorously calibrated, mechanistic framework for understanding how HIV‑1 proteins reshape Fas‑driven apoptosis. By quantifying the shortened survival of re‑activated latent T cells, the study provides a novel metric that could inform the design of eradication strategies aimed at the persistent HIV reservoir.
Comments & Academic Discussion
Loading comments...
Leave a Comment